Indexed In
- Open J Gate
- Cosmos IF
- RefSeek
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- Google Scholar
Useful Links
Share This Page
Journal Flyer
Open Access Journals
- Agri and Aquaculture
- Biochemistry
- Bioinformatics & Systems Biology
- Business & Management
- Chemistry
- Clinical Sciences
- Engineering
- Food & Nutrition
- General Science
- Genetics & Molecular Biology
- Immunology & Microbiology
- Medical Sciences
- Neuroscience & Psychology
- Nursing & Health Care
- Pharmaceutical Sciences
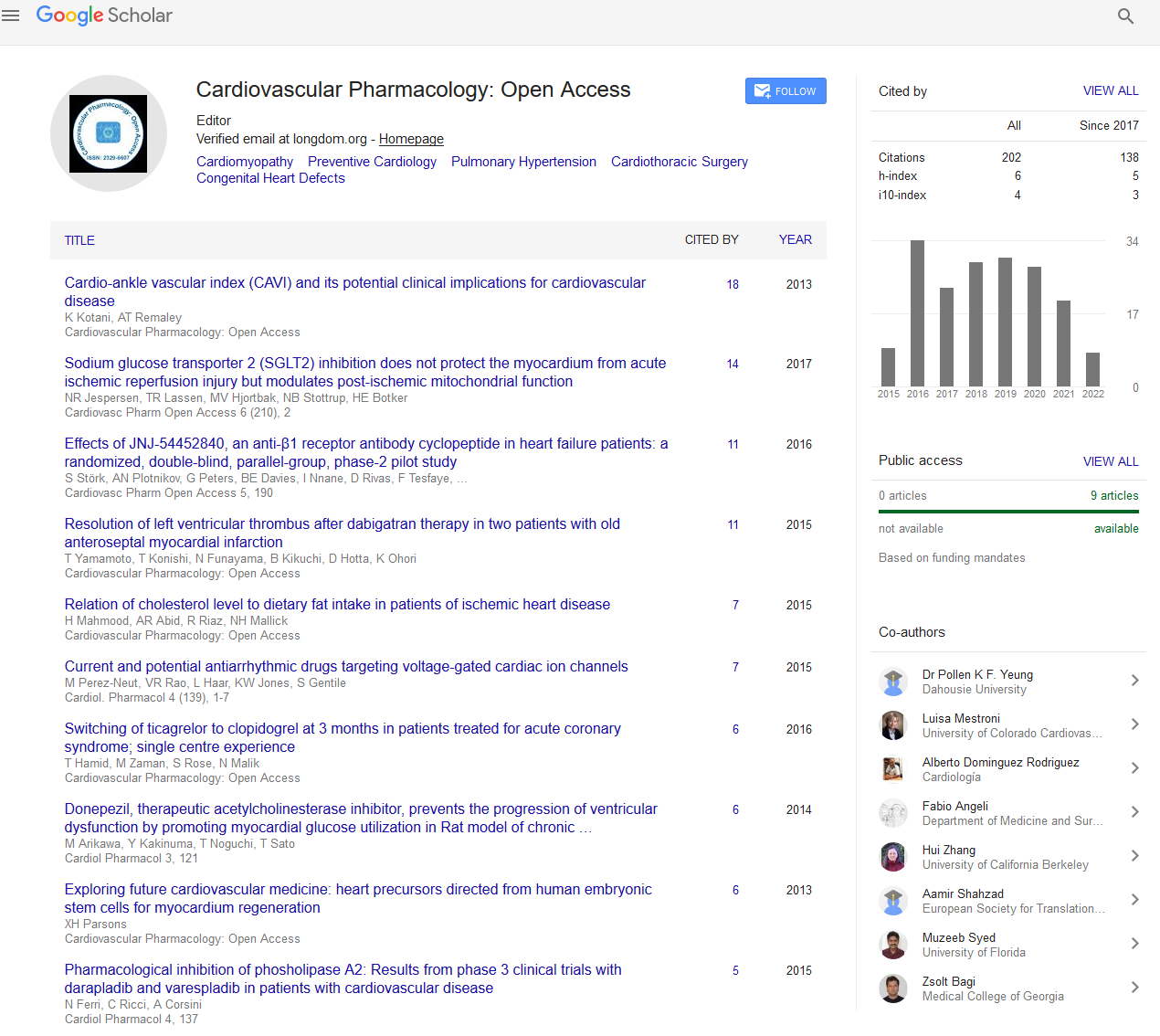
Dexmedetomidine use in patients undergoing electrophysiological study for atrial tachyarrhythmias
11th World Congress on Pediatric Cardiology and Congenital Cardiovascular Disease
April 18-19, 2017 London, UK
Christopher F Tirotta
Nicklaus Children�??s Hospital, USA
Keynote: Cardiovasc Pharm
Abstract:
Dexmedetomidine (DEX) is a selective alpha-2 adrenergic agonist with sedative, analgesic and anxiolytic properties. DEX has not been approved for use in pediatrics. DEX has been reported to depress sinus node and atrioventricular (AV) nodal function in pediatric patients; it was even suggested that the use of DEX may not be desirable during electrophysiological studies (EPS). Other studies have reported no association between DEX use and any significant EKG interval abnormalities in patients with congenital heart disease, other than a decrease in HR. We reviewed all cases presented to the CCL for diagnosis or treatment of atrial tachyarrhythmias since 2007. The patients were stratified into three different groups. Group 1 patients did not receive any DEX. Group 2 patients received a DEX infusion of 0.5-1 mcg/kg/hr. Group 3 patients received a DEX infusion of 0.5-1 mcg/kg/hr and a DEX bolus prior to the infusion of 0.5-1 mcg/kg. We then compared those patients for the following variables: Demographic data and anesthetic data like age, sex, height, weight; mask vs. IV induction, identity of induction agent, amount of sevoflurane and propofol used; amount of DEX used; presence of congenital heart disease and other co-morbidities; the need for isoproterenol (ISO) and dose, the need for adenosine and dose and the need for any other medications to effect rhythm both before and after RFA; the ability to induce the arrhythmia, the type of arrhythmia, the presence of Wolf-Parkinson-White (WPW) syndrome, the presence of an accessory pathway, the ablation rate and the recurrence rate. There were no differences in any of the demographic data between groups. There was no difference in the anesthetic data, except there was a lesser amount of propofol and sevoflurane used in the DEX groups. There was no difference in the electrophysiologic parameters between groups, except the Group 1 patients did require the use of ISO in the pre-ablation period less often compared to the DEX groups. However, there was no difference in the ability to induce the arrhythmia, the percentage of patients ablated, and the recurrence rate between groups.
Biography :
Christopher F Tirotta has been an active member of Miami Children’s Hospital Medical Staff since 1991, practicing in the Department of Anesthesiology. He has served as the Director of Cardiac Anesthesia since 2002. He also works in the Department of Anesthesiology at the University of Miami. He received his BA from Cornell University in 1982 and his MD from New York University School of Medicine in 1986. He also received an MBA degree from Columbia University in 1999. He completed his Internship in Internal Medicine from Stony Brook University in 1987. He completed his Residency training in Anesthesiology from the University of Miami, Jackson Memorial Hospital in 1990. He is sub-specialized in Pediatric and Cardiovascular Anesthesia, including Heart Transplantation. He has been the Principal Investigator for a number of clinical drug/device trials.
Email: christirotta@att.net