Indexed In
- Open J Gate
- Genamics JournalSeek
- JournalTOCs
- Ulrich's Periodicals Directory
- RefSeek
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- Proquest Summons
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- Google Scholar
Useful Links
Share This Page
Journal Flyer

Open Access Journals
- Agri and Aquaculture
- Biochemistry
- Bioinformatics & Systems Biology
- Business & Management
- Chemistry
- Clinical Sciences
- Engineering
- Food & Nutrition
- General Science
- Genetics & Molecular Biology
- Immunology & Microbiology
- Medical Sciences
- Neuroscience & Psychology
- Nursing & Health Care
- Pharmaceutical Sciences
Case Series - (2019) Volume 10, Issue 2
The Many Faces of Thrombotic Microangiopathies
Granfortuna J*Received: 23-Jan-2019 Published: 09-Apr-2019
Abstract
The thrombotic microangiopathies are a complex group of disorders that typically present with a schistocytic hemolytic anemia and associated thrombocytopenia with ensuing microvascular occlusion leading to tissue ischemia and end organ damage. Central nervous system, renal, gastrointestinal and cardiac microcirculations are frequent targets. Signs and symptoms related to organ dysfunction may evolve over weeks to months and may not be present simultaneously. Lactic dehydrogenase enzyme elevation due to microvascular ischemia is frequently disproportionate to elevation of bilirubin or reticulocyte count. The major thrombotic microangiopathies include Thrombotic thrombocytopenic purpura, Disseminated intravascular coagulation/sepsis, and Hemolytic Uremic Syndrome. Hemolytic Uremic Syndrome may be further divided into “typical”, related to Shiga toxin, “atypical”, related to dysregulation or over-activation of Complement, and secondary, including disorders of pregnancy such as the Hepatic enzyme elevation low platelet syndrome or pre-eclampsia, certain other infections such as Streptococccus pneumonia, auto-immune disorders such as Sjogren’s syndrome, cancer, chemotherapy, or other medications, such as quinine and calcineurin inhibitors. These disorders can provoke direct microvascular damage and present as a thrombotic microangiopathy or act as a trigger for a microangiopathic syndrome in individuals with a genetic predisposition. The level of ADAM-TS 13, Von Willebrand Factor cleaving enzyme, is a key discriminator between Thrombotic thrombocytopenic purpura and HUS being severely reduced in Thrombotic thrombocytopenic purpura but not Hemolytic Uremic Syndrome. Plasma exchange with or without steroids is the mainstay of treatment for Thrombotic Thromobocytopenic Purpura. Anti C5 complement antibody therapy has evolved as an important treatment for atypical Hemolytic Uremic Syndrome. Although we have gained significant insight into the pathophysiology of many of these disorders, given the complex interplay between genetic factors, acquired factors, the roles of the humoral, cellular, and innate immune systems, the inflammatory response, and the coagulation system, thrombotic microangiopathies remain clinically challenging. This review will focus on a summary of our current knowledge with regard to diagnosis and treatment of Thrombotic thrombocytopenic purpura and Hemolytic Uremic Syndrome and how they relate to each other and the broader family of thrombotic microangiopathies. Three clinical cases will be used to illustrate key points.
Keywords
Microangiopathy; Schistocyes; Hemolytic anemia; TTP; Thrombotic thrombocytopenic purpura; Hemolytic uremic syndrome; HUS; aHUS; Thrombotic microangiopathy; Complement; Eculizumab
Abbreviations
TTP: Thrombotic Thrombocytopenic Purpura; HUS: Hemolytic Uremic Syndrome; Ahus: Atypical Hemolytic Uremic Syndrome; C’: Complement; LDH: Lactic Dehydrogenase; CNS: Central Nervous System; GI: Gastrointestinal; DIC: Disseminated Intravascular Coagulation; HELLP: Hemolysis Elevated Liver Enzymes Low Platelets; TMA: Thrombotic Microangiopathy; PNH: Paroxysmal Nocturnal Hemoglobinuria; AP Alternative Pathway of Complement Activation; HPF: High Powered Microscope Field
Introduction
The major thrombotic microangiopathies (TMAs) include TTP: Thrombotic Thrombocytopenia Purpura, HUS: Hemolytic Uremic Syndrome, and DIC/Sepsis. HUS is further subdivided into Shiga toxin related (STEC HUS) provoked by pathogenic strains of E. coli 0157:H7 and 0104:H4, and Atypical HUS (aHUS). aHUS can be further subdivided into primary disorders of complement regulation, and HUS resulting from secondary disruptions of the complement system or direct vascular endothelial damage not clearly related to complement such as drug/toxin mediated with considerable clinical overlap between the two (Table 1). A number of conditions may result in or trigger a TMA (Table 2). TMA syndromes nature vs nurture TMA syndromes can be hereditary, acquired, or both: (Tables 1 and 2) but a common thread is an initial insult to the vascular endothelium [1-5]. When a patient presents with a COOMB’s negative schistocytic hemolytic anemia with associated thrombocytopenia, the first consideration is whether the process is TTP, HUS, or another secondary cause of microangiopathy. Three clinical vignettes will be used to illustrate the variable presentation and course of these disorders.
| Hereditary | Acquired |
|---|---|
| Adam-TS-13 Deficiency: Upshaw Schulman Mutations in Complement regulatory proteins Mutations in genes for methylmalonic acid and homocysteine (Cobalamine deficiency with normal B12 levels)-rare Mutations in diacylglycerol kinase (DGKE), congenital recessive aHUS: complement activation/thrombosis; AKI,-rare |
Auto-antibodies to ADAM-TS-common Antibodies to C’ regulatory proteins-rare Shiga Toxin related HUS Other infections: Strep Pneumoniae; Influenza Drug mediated immune ex quinine Drug mediated toxic ex oxymorphone LA |
Table 1: Common conditions associated with thrombotic microangiopathy [1]. Both hereditary and acquired factors may lead to the development of a thrombotic microangiopathy. The common pathogenetic mechanism is an insult to the vascular endothelium.
| Systemic Infection |
| Malignant Hypertension |
| Pregnancy: HELLP syndrome; Pre-eclampsia |
| Autoimmune disease: Lupus; Scleroderma renal crisis; Vasculitis |
| Catastrophic Antiphospholipid Antibody Syndrome |
| Cancer: Gastric, Pancreatic, Breast, Mucin secreting adenocarcinomas |
| Chemotherapy: Mitomycin C, Gemcitabine, Bevacizumab |
| Other Drugs: Calineurin inhibitors, Extended release opioids, cocaine, quinine, ticlopidine, Clopidigrel, Interferon |
| Organ and Bone marrow transplant |
Table 2: Secondary conditions that may result in or trigger a thrombotic micoangiopathy.
Case Series
Clinical vignette 1
A 37-year-old African American man in previous good health presented to the emergency department with the acute onset of right flank and abdominal pain. There was no history of trauma. He was not on anticoagulants or anti-platelet agents. Six months ago he went to an urgent care center and was treated for Streptococcus pharyngitis; he was told his blood pressure was elevated.
He denied alcohol, tobacco, or drug use except for occasional marijuana use. On exam: Blood pressure was 192/134, optic discs sharp; no retinal hemorrhages or exudates. No focal neurologic deficits. Tender on palpation over the right flank.
• EKG: sinus rhythm, left ventricular hypertrophy with strain pattern.
• CT abdomen: large right peri-nephric hemorrhage.
• Lab: hemoglobin 12 grams, Hematocrit 38%, platelet count 128,000, fell to 106,000 over next 24 hours; random glucose 269; BUN 29, creatinine 3.2; repeat in 24 hours: BUN 45, creatinine 5.1
• Bilirubin 0.3, reticulocyte count 1.8%, Haptoglobin 45 mg, prothrombin time 14.1 seconds, COOMB's test negative.
• Urine: large positive for hemoglobin and protein. No casts.
• Review of the peripheral blood film with 1-2 schistocytes per high powered field.
Clinical vignette 2
A 58-year-old African American man with prior history of alcohol abuse initially presented to the emergency department with chest pain. Blood pressure 166/92; Exam otherwise normal; No acute changes on cardiogram; Hemoglobin 17.5; WBC 3,800; platelets 107,000; BUN 15, creatinine 1.2.
He was started on lisinopril and discharged. He returned 4 days later complaining of leg weakness and dizziness. BP 210/100; CT of the head showed an acute left thalamic infarct; Troponin 1.05.
He was admitted with the diagnosis of a hypertensive stroke and a non ST wave elevation MI. Left ventricular ejection fraction 50-55% on echocardiogram. He had a high risk myoview nuclear medicine stress test but coronary catheterization showed normal coronaries.
Post hydration labs with Hemoglobin 14. White counts in range of 3,800-5,000, platelets 71,000-115,000, normal liver chemistries. Hypertension treated and he was discharged.
• He continued to have intermittent chest and abdominal pain over the next 3 months.
• He presented to the hospital again with complaints of diffuse abdominal pain. Abdominal exam was unremarkable and a CT scan of the abdomen was normal. Stool was negative for occult blood.
• Hemoglobin 9.6, WBC 14,000; platelets 116,000; New liver chemistry abnormalities with SGOT 181, SGPT 107, Bilirubin 1.3, alkaline phosphatase 249.
• He was discharged from the emergency department only to return 4 days later when he was found outside in the rain wandering in a delirious state.
• Exam: delirious, agitated, not following commands or verbally communicative, PERRL, moving all extremities, no facial asymmetry, symmetric reflexes. BP 128/103, pulse 102 regular, respirations 38/minute, Temp 97.4, oxygen saturation 95% on room air. No cardiac murmur. Abdomen nontender; No ecchymoses.
• Lab: Hemoglobin: 8.1, platelets 15,000, BUN 37, Creatinine 2.1, SGOT 212, SGPT 223, alkaline phosphatase 512, LDH 2,345, bilirubin 3.3, reticulocyte count 1.8%, Troponin 6.5, peak to 19.7, urine large positive for Hemoglobin and bilirubin.
• COOMB's direct and indirect negative, Prothrombin time 13.7 seconds, PTT 25 seconds
• Review of peripheral blood with 1-3 schistocytes per HPF, presence of microspherocytes, less than 1 platelet per HPF
• CT brain: no new abnormalities. CT abdomen and pelvis showed no focal abnormalities of the liver or biliary tract.
Discussion
There have been two fundamental advances in our thinking about TMAs: first, understanding the critical role of Ultra-large Von Willebrand multimers and the importance of the von Willebrand Factor cleaving enzyme ADAM-TS (a disintegrin and metalloproteinase with thrombospondin type 1 repeats, member 13) in the pathogenesis of TTP and second, is the role that Complement regulation or dysregulation plays in the development of the microangiopathic process in aHUS and its variants [2-12].
TTP epidemiology
TTP has an annual incidence of 10 cases per million population in the US. 90% of cases occur in adults; 10% in children and adolescents. Etiology is primarily autoimmune due to antibodies against the ADAM-TS enzyme.
Rarely, TTP occurs as a hereditary form: Upshaw Schulman Syndrome. It is twice as common in women. There is an increased incidence in African Americans. There is a high tendency to relapse and a 10-20% mortality rate despite contemporary treatment.
TTP pathophysiology
Severe acquired or inherited deficiency of ADAM-TS 13 Von Willebrand factor cleaving enzyme results in increased adhesion of platelets to ultra-large Von Willebrand multimers leading to the formation of platelet rich microthrombi within small arterioles (Figure 1).
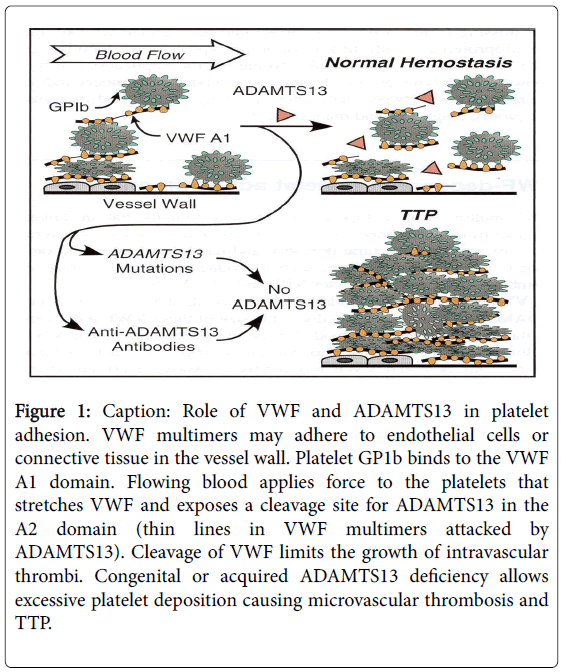
Figure 1: Caption: Role of VWF and ADAMTS13 in platelet adhesion. VWF multimers may adhere to endothelial cells or connective tissue in the vessel wall. Platelet GP1b binds to the VWF A1 domain. Flowing blood applies force to the platelets that stretches VWF and exposes a cleavage site for ADAMTS13 in the A2 domain (thin lines in VWF multimers attacked by ADAMTS13). Cleavage of VWF limits the growth of intravascular thrombi. Congenital or acquired ADAMTS13 deficiency allows excessive platelet deposition causing microvascular thrombosis and TTP.
There is minimal or no acute inflammatory response in the vessel wall as opposed to complement mediated processes; there is no recruitment of inflammatory cells or fibrin deposition; there is the formation of a primarily bland platelet “white” thrombus (Figure 2).
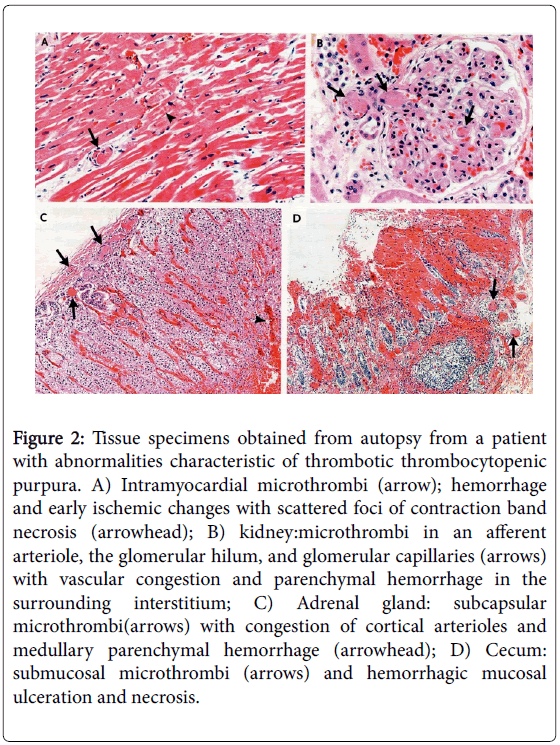
Figure 2: Tissue specimens obtained from autopsy from a patient with abnormalities characteristic of thrombotic thrombocytopenic purpura. A) Intramyocardial microthrombi (arrow); hemorrhage and early ischemic changes with scattered foci of contraction band necrosis (arrowhead); B) kidney:microthrombi in an afferent arteriole, the glomerular hilum, and glomerular capillaries (arrows) with vascular congestion and parenchymal hemorrhage in the surrounding interstitium; C) Adrenal gland: subcapsular microthrombi(arrows) with congestion of cortical arterioles and medullary parenchymal hemorrhage (arrowhead); D) Cecum: submucosal microthrombi (arrows) and hemorrhagic mucosal ulceration and necrosis.
TTP clinical features
The sine qua none of the diagnosis is the presence of a COOMB’s negative microangiopathic hemolytic anemia with severe thrombocytopenia. The classic pentad which also includes fever, confusion, and renal failure is only present in 10% of patients at presentation but about 60% of patients do have neurologic signs initially. Acute renal failure is unusual and creatinine is typically less than 2. Not commonly recognized is both cardiac and mesenteric ischemia occurring in 25% and 35% of patients respectively. Symptoms typically evolve over time. There may be a prodrome of atypical chest or abdominal pain antedating the diagnosis by weeks to months (personal observations; cf case 2). Fever is not uncommonly associated with hemolytic anemia and likely reflects a toxic inflammatory effect of free hemoglobin and not an underlying infection in most instances [13-16]. Clinical presentation can be quite variable and can occur in association with: pregnancy: pre-eclampsia, eclampsia, HELLP syndrome (hemolysis elevated liver enzymes, low platelets); Drugs: quinine; ticlopidine; clopidogrel, Opana long acting morphine, calcineurin inhibitors; Chemotherapy agents: including mitomycin C, Gemcitabine; Biologic agents: including bevacizumab and interferon; Malignancy; Infection: HIV, CMV, EBV; Post Bone marrow transplant. Two rare metabolic syndromes have been described: Cobalamin C defect TMA caused by mutations in the gene encoding the methyl malonic aciduria and homocystinuria type C protein (MMACHC) and Diacylglycerol kinase epsilon (DGKE) deficiency TMA. Most cases of Cobalamin C defect TMA occur in neonates although rare cases have been reported in adults. Plasma levels of methylmalonic acid and homocysteine are elevated but B12 levels are typically normal. Treatment is high dose parenteral vitamin B. DGKE mutation TMA usually presents in the first year of life with hematuria, hypertension, and proteinuria. The mechanism of TMA remains unclear and treatment is supportive with no role for plasma exchange or anticomplement therapy. In an in vitro model, Bruneau, et al. show that disruption of DGKE does not upregulate complement but appears to induce endothelial damage via activation of the p38 MAP kinase pathway. This may be a novel mechanism for endothelial damage resulting in a TMA [16-30].
TTP laboratory features
Schistocytes are seen on the peripheral blood film. Microspherocyes are also a common finding and are rarely seen under other circumstances except for acute burn injury. Thrombocytopenia is usually severe, less than 30,000, compared with HUS where thrombocytopenia is more moderate. LDH is typically disproportionately elevated compared with bilirubin and reticulocytes reflecting microvascular ischemia [16,31,32]. PT and PTT and fibrinogen are typically normal which helps to distinguish from DIC but d-dimer may be elevated. There may be a modest elevation of creatinine with or without proteinuria or hematuria. Notably, troponin is elevated in up to 60% of patients with EKG changes seen in 10% likely reflecting ischemia in the cardiac microcirculation [16].
ADAM-TS enzyme is typically less than 5%-10% of control values. Severely decreased ADAM-TS enzyme level is the sine qua non that results in TTP.
TTP treatment
Plasma exchange with 1-1.5 plasma volumes daily until platelet count is >/=150,000 for 2 successive days is the current standard of care. Some experts then favor a tapering plasma exchange schedule for 1-2 weeks after obtaining a complete response although this has not been systematicly studied. Steroids in variable doses of 1-10 mg/kg/day of solumedrol or equivalent dose of prednisone are routinely used in view of the autoimmune nature of most cases.
Pre-plasma exchange steroid dosing also serves to mitigate minor transfusion reactions due to large volumes of plasma administered. Rituximab monoclonal anti CD 20 antibody has been used in the relapsed refractory setting; benefit as part of initial therapy has also been reported [15]. Pre-emptive use of Rituxan in patients considered at high risk for relapse based on persistent decreased ADAM-TS enzyme levels has been reported to improve durable remission rates in one third of patients at risk [33]. Splenectomy and/or immunosuppressive drugs such as Cytoxan, vincristine, Azathioprine, or cyclosporine have shown responses in refractory disease [1,13,15,34-48].
TTP novel therapies
Caplacizumab, an anti Von Willebrand factor receptor nanobody derived from the llama, which prevents binding of VWF to platelet glycoprotein Ib, has improved time to response and decreased the number of plasma exchanges to achieve complete response in a phase II clinical trial. These observations were confirmed and extended in a phase III trial completed in 2017 which has led to FDA approval [15,16,49-51]. Following a 10 mg IV loading dose given before the first planned plasma exchange then daily subcutaneously during plasma exchange then for 30 days, median time to normalization of platelet count was shorter but more importantly, the composite outcome of recurrence of TTP, TTP related death, or a thromboembolic event during treatment, was markedly improved at the expense of a moderate 17% increase in nonfatal bleeding [52]. Addition of this new modality is poised to become a new standard of care used in conjunction with plasma exchange. How to best integrate this expensive new drug with other agents, in particular, Rituximab, will need to be studied prospectively.
N-Acetylcysteine (mucormist) has shown activity and it’s use is currently being explored. This drug breaks the disulfide bonds that hold together Von Willebrand multimers. Shorter multimers are postulated to decrease adhesion of platelets to the vascular endothelium [53-56]. The role of recombinant ADAM-TS enzyme is being explored [57]. It’s main use may be limited to the congenital form of TTP although one study demonstrated the ability of recombinant ADAM-TS was able to override inhibitory antibodies in the plasma of patients with acquired TTP [58]. Proteosome inhibitors such as Bortezomib (Velcade) have shown activity in relapsed TTP. The mechanism of action is felt to be depletion of plasma cell producing anti ADAM-TS enzyme antibodies [40].
Table 3 summarizes available treatments for TTP.
| Primary Treatment: |
| Plasma exchange plus corticosteriods |
| Rituximab anti CD-20 Antibody |
| Caplacizumab anti GP 1b Antibody |
| Relapsed/Refactory: |
| Rituximab |
| Immunosuppressive agents; bortezomib; cyclosporine; cyclophosphamide; vincristine |
| Recombinant ADAM-TS enzyme replacement |
| N-acetyl Cysteine |
| Splenectomy |
Table 3: Plasma exchange serves to remove antibodies against ADAMTS and simultaneously replenish this critical enzyme in the pathogenesis of TTP. Steroids downregulate abnormal antibody production and Rituximab selectively removes anti CD-20 antibody producing cells. By inhibiting Von Willebrand-platelet interactions, caplacizumab improves time to platelet response and decreases morbidity when added to the initial treatment of TTP. N-acetyl cysteine can break disulfide bonds between VW multimers inhibiting platelet adherence to endothelial cell anchored ultralarge VWF multimers.
TTP prognosis
80%-90% of patients survive after treatment of the initial episode. 40% of patients have one or more relapses. HIV infected patients have similar response rates as non HIV infected patients. Fetal extraction does not correct TTP in contrast to pre-eclampsia and HELLP syndrome so pregnancy need not routinely be interrupted. ADAM-TS levels may not be a good predictor of relapse. There is an increased risk for developing a collagen vascular disease. There are increased rates of neuro-cognitive deficits, arterial hypertension, and major depression after recovery of blood counts. Very high LDH, over 10 times upper limit of normal, and troponin levels over 0.25, portend a poor prognosis with increased chance of refractoriness to treatment and death [14-16].
Clinical course patient 1
Patient presenting with a hypertensive crisis, a spontaneous retroperitoneal bleed, rapidly worsening renal failure, moderate thrombocytopenia, elevated LDH, and the presence of schistocytes on peripheral blood film. Microangiopathy related to malignant hypertension vs early TTP vs aHUS? Aggressive treatment of hypertension initiated with parenteral agents. In view of rising creatinine and LDH and falling platelet count, a vascular catheter was placed. 1 volume daily plasma exchange initiated. ADAM-TS enzyme returned at 61% of control (68-163). As blood pressure came under control, renal function stabilized, LDH fell, and platelet count rose to normal. Plasma exchange stopped after 3 treatments. Creatinine improved from peak 4.5 to 2.8 by day 24.
This patient was felt to have microangiopathy related to malignant hypertension. It is postulated that by activating the renin-angiotensinaldosterone system and causing increased shear forces in the microvasculature, malignant hypertension contributes to endothelial injury and promotes microangiopathy mimicking TTP [59-62].
Clinical course patient 2
Patient with a 3 month prodrome of atypical chest and abdominal pain, non ST elevation MI with significant elevation of cardiac troponin but normal coronaries, a thalamic stroke, subacute abdominal pain and abnormal liver function tests with negative abdominal imaging, who then had progressive neurologic deterioration and an acute fall in hemoglobin and platelets. The patient required intubation to protect his airway. A vascular catheter was placed and plasma exchange and steroids were initiated. He had a rapid, dramatic, improvement in his mental status, rise in platelet count to 344,000, fall in LDH to 228, and normalization of renal function. A total of 9 plasma exchanges done. ADAM-TS enzyme returned at <10% of control solidifying the diagnosis of TTP. This case illustrates that there may be a prolonged prodrome before the diagnosis is established. It is likely that the fluctuating cardiac and GI symptoms are the consequence of platelet thrombi to the coronary and mesenteric microcirculations respectively. Marked elevation of serum LDH with concomitant new liver function abnormalities in absence of any focal abnormalities of the liver or biliary system on CT scan probably reflects microvascular injury to the liver in addition to hemolysis.
This patient went on to have 2 subsequent relapses successfully reinduced into remission with plasma exchange and steroids. Azathioprine added following the second relapse with no subsequent recurrences out over 2 years.
Hemolytic Uremic Syndromes
Clinical vignette 3
57-year-old African American male diagnosed with Large Granular T Cell Leukemia 6 years ago treated with initial observation then 2 years ago received chemotherapy with weekly cladribine for 3 months. Baseline chronic renal insufficiency with creatinine in range 1.5-1.8. Initially admitted with cough, fever, dyspnea; bibasilar and right middle lobe infiltrates on chest X-ray. Positive Streptococcus Pneumonia antigen. He was treated with Ceftriaxone and Azithromycin then transitioned to oral Augmentin.
• Initial BUN 47, creatinine 2.8. Creatinine 1.4 at discharge.
• Initial platelet count 220,000, fell to 77,000, rose to 144,000 by discharge.
• He presented 7 days later with persistent cough and progressive dyspnea.
• Exam: thin male dyspneic at rest. BP 174/108, pulse 106 regular, respirations 22, Temp 98.8
• Scattered expiratory wheezes over the lung, regular cardiac rhythm, no murmur, gallop, or rub, no adenopathy, no organomegaly, no focal neurologic deficits.
• CXR: pulmonary infiltrates improved but not completely resolved.
• BUN 118, creatinine 15.8, 3+ proteinuria, microscopic hematuria, 6-30 red cells, granular casts.
• Urine drug screen positive for cocaine.
• Hemoglobin 8, Platelet count initial 240,000, LDH 347, bilirubin 0.8.
• COOMB's negative, Haptoglobin 2 weeks prior 349.
• Initial blood film review with numerous platelets, 1+ spherocytes, no polychromasia, rare, <1/10 HPF, schistocytes.
The hemolytic uremic syndromes are thrombotic microangiopathies characterized by predominant renal failure. They include toxic/ infectious causes which are not directly complement mediated and are termed “typical” and inherited or acquired conditions in which they are directly complement mediated, “Atypical HUS”, or indirectly complement mediated. It would seem for the adult population “Atypical HUS” is a misnomer since complement mediated disorders are far more common [1,9,10,16,17,46-49].
STEC shiga toxin related HUS
STEC HUS is more common in children under 5 with an incidence of 6.1 cases per 100,000 children compared with 1-2/100,000 in adults. Patients typically, but not always, present with bloody diarrhea. E Coli subtypes 0157:H7 and 0111:H8 predominate. These enterohemorrhagic bacteria adhere to the colonic mucosa which they invade and replicate in causing cell destruction. The Shiga toxin binds with high affinity to terminal galactose disaccharides in globotriaosylceramide (glycolipid Gb3) receptors in the membranes of colonic, cerebral, renal, glomerular, mesangial, and tubular cells, monocytes, and platelets resulting in cytokine release, release of large VWF multimers, platelet aggregation, and inhibition of protein synthesis leading to cell death. The high expression of Shiga toxin binding glycolipid receptors on renal cells accounts for the high incidence of acute kidney injury. Although there is an increase in high molecular weight Von Willebrand multimers, there is only a mild to moderate decrease in ADAM-TS enzyme levels.
Although severe, SHIGA related HUS is usually self-limited. There is no defined role for plasma exchange at this time but there may be a subset of patients presenting with severe disease who may benefit and this is the subject of current clinical trials. Additional infection related mechanisms of cell death include the production of neuraminidase by invasive pneumococcal infection or the influenza virus which can expose the Thomson-Friedenreich surface antigen by removing sialic acid residues on red cells and platelets and provoke an IgM antibody response causing complement mediated damage, a positive C’3 complement direct COOMB’s test, and can result in a high mortality rate [9-11,32,33,63-67].
Atypical hemolytic uremic syndrome (aHUS)
aHUS is a manifestation of over-activation or dysregulation of complement resulting in vascular endothelial damage. aHUS is likely triggered by a precipitating event in a genetically susceptible host in the majority of cases. Glomerular endothelial cells are particularly vulnerable to complement mediated damage with about 50% of patients developing end stage renal disease. Platelet activation from endothelial damage as well as complement dysregulation on the platelet surface leads to a pro-thrombotic state. Additional proinflammatory and pro-thrombotic responses occur with chemotaxis of myeloid cells, formation of neutrophil extracellular traps (NETs) and degranulation of mast cells [49].
Innate immunity and the complement system
The complement system plays a major role in innate immunity. The classic and Lectin complement pathways depend on antigen-antibody opsonization of pathogens and subsequent removal by the reticuloendothelial system. In contrast, the Alternative Complement Pathway (AP) is constitutively active as a result of spontaneous hydrolysis of C3 to C3b. C3b acts as a protective shield which is deposited on the surfaces of red cells, white cells, and platelets. The special feature of the alternative pathway is the ability to spontaneously attack any surface to defend against any biomaterial, such as a bacterial cell wall. All that is required is a permissive surface and not antigenantibody recognition. The downside is potential attack of the host cell as an innocent bystander with subsequent collateral cell damage. This makes continuous downregulation of activation essential. This is where factor H, the plasma regulator of the AP, plays a central role in determining whether a target cell is spared or destroyed. If factor H binds to C3b, activation is inhibited. In the absence of binding, activation to the anaphylotoxin C5a occurs, ultimately activating the rest of the complement cascade leading to the formation of the membrane attack complex and cell destruction (Figure 3) [10-12,63,65,68,69].
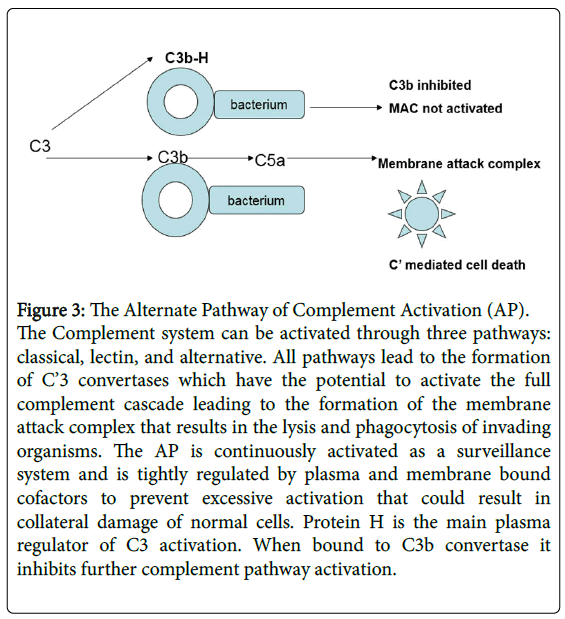
Figure 3: The Alternate Pathway of Complement Activation (AP).
The Complement system can be activated through three pathways: classical, lectin, and alternative. All pathways lead to the formation of C’3 convertases which have the potential to activate the full complement cascade leading to the formation of the membrane attack complex that results in the lysis and phagocytosis of invading organisms. The AP is continuously activated as a surveillance system and is tightly regulated by plasma and membrane bound cofactors to prevent excessive activation that could result in collateral damage of normal cells. Protein H is the main plasma regulator of C3 activation. When bound to C3b convertase it inhibits further complement pathway activation.
aHUS: The role of complement
aHUS is due to uncontrolled activation of the Alternative Pathway of Complement in about 60% of cases. The complement system is controlled by a combination of plasma factors: factors H, I, B, and membrane factors: MCP (membrane cofactor protein, CD46, CD55, and CD59) that have complex interactions in inhibiting or promoting the formation of the terminal complement cascade with the potential formation of the membrane attack complex. Dysregulation of this pathway can occur by loss of function of regulatory proteins leading to endothelial injury, platelet activation, and thrombosis. A number of regulatory genes have been shown to be mutated in aHUS. Factor H is the most important regulator. Mutations in factor H are responsible for 20-60% of all aHUS cases. Glomerular endothelial cells have high expression of Factor H receptors and are particularly sensitive to complement mediated damage.
MCP, membrane cofactor protein, also plays a major role in regulating complement activation and is also highly expressed on glomerular endothelial cells. In view of the fact that it is a membrane bound protein, it cannot be removed by plasma exchange (Figure 4).
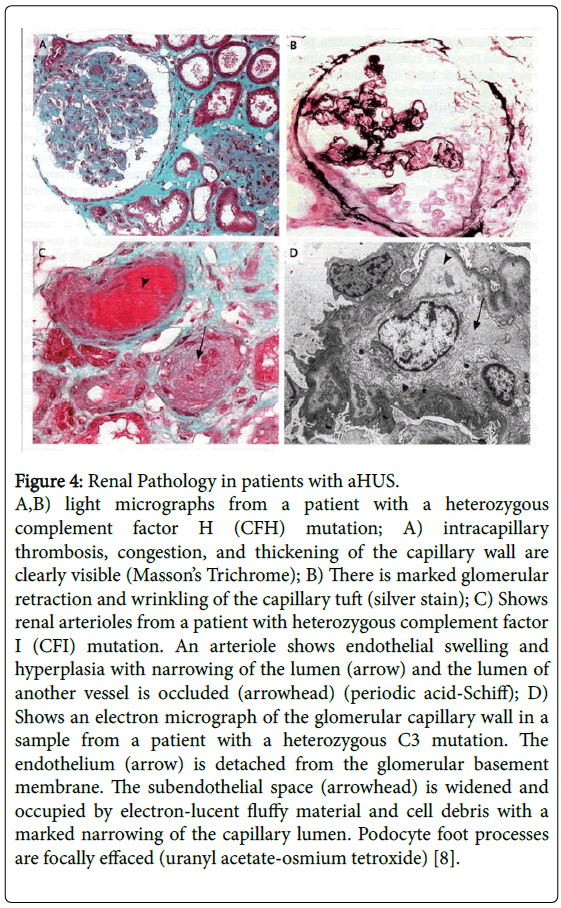
Figure 4: Renal Pathology in patients with aHUS.
A,B) light micrographs from a patient with a heterozygous complement factor H (CFH) mutation; A) intracapillary thrombosis, congestion, and thickening of the capillary wall are clearly visible (Masson’s Trichrome); B) There is marked glomerular retraction and wrinkling of the capillary tuft (silver stain); C) Shows renal arterioles from a patient with heterozygous complement factor I (CFI) mutation. An arteriole shows endothelial swelling and hyperplasia with narrowing of the lumen (arrow) and the lumen of another vessel is occluded (arrowhead) (periodic acid-Schiff); D) Shows an electron micrograph of the glomerular capillary wall in a sample from a patient with a heterozygous C3 mutation. The endothelium (arrow) is detached from the glomerular basement membrane. The subendothelial space (arrowhead) is widened and occupied by electron-lucent fluffy material and cell debris with a marked narrowing of the capillary lumen. Podocyte foot processes are focally effaced (uranyl acetate-osmium tetroxide) [8].
Any complement activating condition can unmask aHUS. There is a delicate balance between downregulation and activation of the system which becomes perturbed in a number of physiologic and pathologic conditions There is considerable overlap in the conditions that may also result in TTP. Both conditions result in an insult to the vascular endothelium but the pathogenesis differs. The virtual absence of ADAM-TS enzyme in TTP results in a non-inflammatory platelet thrombus as opposed to the inflammatory vascular changes associated with complement mediated vascular damage (Table 4) [1,9,10,19,32,63].
| Mutations in Complement regulatory proteins |
| Autoantibodies against factor H |
| Malignant Hypertension |
| Pregnancy and postpartum |
| Infection |
| Surgery/Trauma |
| Medication/Drugs |
| Post organ Transplant |
| Autoimmune Conditions |
| Malignancy |
Table 4: Complement activating conditions that can activate or unmask aHUS.
More than 50 mutations in complement regulatory proteins have been described. (Table 5). Their presence is necessary but not always sufficient to result in clinical aHUS and aHUS can occur in the absence of a previously defined mutation. The clinical expression and severity of the microangiopathic process may reflect the particular mutation. As stated, up to 60% of the mutations found in aHUS occur in the factor H gene. Mutations in the MCP gene occur in 5%-9% of patients and are associated with a milder clinical course with only 6%-19% progressing to end stage renal disease. This subset is not responsive to plasma exchange. Mutations in C3 complement, Factor B, occur in 1%-2% of patients and result in hyperactivation of the alternative pathway and are associated with a more aggressive clinical course with over 88% of patients progressing to ESRD.
| Form of Disease | Complement Abnormalities |
|---|---|
| Familial | Mutations in CFH, 40-45%; in CFI, 5-10%; in C3, 8-10%; in MCP, 7-15%; in THBD, 9%; and in CFB, 1-2% |
| Sporadic idiopathic | Mutations in CFH, 15-20%; in CFI, 3-6%; in C3, 4-6%; in MCP, 6-10%; in THBD, 2%; and in CFB, 2 cases; anti-CFH antibodies: 6-10% |
| Pregnancy-associated | Mutations in CFH, 20%; in CFI, 15% |
| HELLP syndrome | Mutations in CFH, 10%; in CFI, 20% and in MCP, 10% |
| Drugs | Rare CFH mutations (Mostly unknown) |
| Organ transplantation | Mutations in CFH, 15%; in CFI, 16% |
| Human immunodeficiency virus infection | Unknown |
| Cancer | Unknown |
Table 5: Classification of Atypical Hemolytic Uremic Syndrome [57]. More than 50 mutations in complement regulatory proteins have been described.
Testing for mutations is generally not helpful in the acute situation except if antibodies to factor H are detected. In that case, plasma exchange in addition to immunosuppressive agents such as steroids, Rituxan, or cyclophosphamide is the preferred treatment. Mutational testing is available in specialty laboratories if indicated (http://machaondiagnostics.com and others) [7,8,70].
aHUS clinical features
In addition to the usual signs and symptoms that might accompany severe anemia or thrombocytopenia, patients may present with new onset or acutely worsening hypertension from renovascular compromise. Signs of acute renal injury may include edema, proteinuria, and hematuria. Extra-renal manifestations occur in 20%-25% and can include cardiovascular, i.e chest pain; neurologic, i.e. change in mental status, focal neurologic deficits, or seizures; GI, i.e. abdominal pain, vomiting, diarrhea, pancreatitis, or hepatitis.
Concomitant infection is not uncommon. When occurring during pregnancy, aHUS typically, but not exclusively, presents postpartum compared with ante-partum in TTP. There is a complex relationship between preeclampsia and the Hemolysis, elevated liver enzymes, low platelet syndrome (HELLP). The HELLP syndrome may be an extreme manifestation of preeclampsia/eclampsia but can occur in about 20% of women in the absence of hypertension or proteinuria. Based on the observation that the HELLP syndrome is a systemic inflammatory disorder and on findings that mutations in complement regulatory proteins occur in some women with preeclampsia, a woman with severe, early, HELLP syndrome was treated with the anti C5a complement drug eculizumab with marked improvement suggesting that this drug may be an important component of management in some of these women [10,14,18,54,58,71-73].
aHUS diagnosis and treatment
After documentation of a COOMB’s negative* microangiopathic hemolytic anemia and associated thrombocytopenia with presence of schistocytes on the peripheral blood film and an elevated serum LDH it is important to document involvement of at least one organ system: renal, GI, neurologic, or lung (aHUS but not TTP can affect the lungs and may help distinguish between the two); investigate any diarrhea: obtain a rectal swab for Shiga toxin by PCR or ELISA, stool and urine cultures. (Note: GI symptoms alone cannot distinguish between STECHUS, aHUS, or TTP); begin treatment of any obvious co-existing disease or infection; and obtain a pre-treatment ADAM-TS enzyme level. Eculizumab anti-C5a monoclonal antibody is the current treatment of choice but plasma exchange could be initiated if the diagnosis of aHUS is not secure at the time of presentation in an unstable patient pending results of ADAM-TS enzyme analysis [1,10,62,64].
If ADAM-TS enzyme is >/= 10% of control from a reliable laboratory, discontinue plasma exchange, administer triple vaccines against Pneumococcus, Meningiococcus, and Hemophilus Influenzae, start a prophylactic course of oral broad spectrum antibiotics then begin a trial of Eculizumab anti-C5a antibody. There appears to be an ongoing risk for infections with encapsulated organisms, in particular Meningiococci, while on eculizumab and most authorities recommend continuation of prophylactic antibiotic therapy for the duration of treatment.
Renal, skin, or gingival biopsy may be indicated in rare instances to establish the diagnosis.
*Complement COOMB’s may be positive in association with IgM mediated antibody response to certain infections. See text.
Eculizumab
Eculizumab is a humanized monoclonal antibody to C’5a complement which prevents the formation of the membrane attack complex. It’s first successful use was in the treatment of PNH, a complement mediated stem cell disorder resulting in non-immune hemolysis [69,74].
It has revolutionized the treatment of patients with complement mediated aHUS. Clinical trials have shown protection against further renal damage and, in some instances, resolution of severe renal damage/dialysis dependence. This has not been the case in patients with aHUS treated with plasma exchange alone where there is often progressive renal dysfunction despite a hematologic response [10-12,14,44-46,48,52,57-61].
It may take a long time for an over-activated complement system to become quiescent. Once started, the optimal duration of treatment is unclear. In the original prospective registration trials, median duration of treatment was 60-64 weeks with a range of up to 145 weeks [75,76]. In a trial of pediatric patients, median duration of treatment was 2 years. In that study there was a trend for higher relapse rates in patients with CFH mutations compared with patients with CFI and MCP mutations or no mutations [77]. In a recent review, the authors recommend a minimum of 6 month of treatment after normalization or stabilization of renal function. In a series of 17 patients treated at Johns Hopkins University, median duration of treatment was 90.5 days with range 15-545. Three patients relapsed, consistent with previous observations that about 30% of patients relapse when treatment is discontinued. Mutational and antibody testing did not appear to guide optimal time to stop therapy and available evidence at the time of their report suggests TMA manifestations following discontinuation are unpredictable”. Efforts are in progress to develop parameters that would guide the safe discontinuation of the drug including measurement of eculizumab trough levels as an indicator of optimal therapy, assays of CH50 levels to assess adequacy of complement blockade, and mutational analysis of C5 complement in cases of eculizumab resistance. A detailed approach for the use and monitoring of eculizumab therapy, and an algorithm to assess low versus high risk situations to guide discontinuation of the drug is provided in a recent excellent review by Fakhouri and Loirat [10-12,32,62-64,66,69,76,78-81].
TTP vs HUS: How are they the same? How are they different?
The pathogenesis of TTP is primarily related to vascular occlusion by platelets whereas the pathogenesis of other primary TMA syndromes also includes injury to vascular endothelial and renal cells with an associated inflammatory response. The thrombus in TTP is primarily composed of platelets “white thrombus”; in HUS, the thrombus contains platelets, fibrin, and inflammatory cells “red clot”.
ADAM-TS enzyme may be decreased in aHUS but usually never less than 10%. The profound deficiency of ADAM-TS enzyme in TTP compared with the more modest decrease in aHUS provides the rationale for the use of plasma exchange/plasma infusion in TTP to simultaneously remove autoantibodies in the acquired form, and to replenish the enzyme in both hereditary and acquired forms. Although there may be a moderate consumption of ADAM-TS in some cases of aHUS, except for cases where the pathogenesis is related to anti-factor H antibodies, inhibition of an over-activated complement system and not plasma exchange is the mainstay of treatment.
About 20% of aHUS patients can have normal renal function on initial presentation but most progress to significant renal dysfunction whereas most TTP patient have only mild renal impairment which usually does not progress.
Schistocytes may be rare, <1 per high power microscope field, initially with both TTP and HUS but may increase in number as the disease process evolves, therefore, serial monitoring of blood smears is important. The presence of microspherocytes may be an additional finding that helps establish the diagnosis of TTP or HUS when schistocytes are rare (personal observations and communications with Dr J George). The platelet count is typically only moderately decreased or initially normal with aHUS but typically severely decreased, less than 30,000 with TTP. The lung can be a target organ in HUS but not TTP.
Cross-talk
The complement system has widespread interactions with the acute inflammatory response system and the coagulation system with intriguing roles in human pathophysiology. In PNH, a mutation in the PIG-A gene results in decreased expression of two complement control proteins, CD55 and CD59. As a result, red blood cells are more sensitive to complement mediated lysis. This is not a microvascular insult. There is no endothelial damage and no schistocytes are seen. There is a strong association with large vessel thrombosis but not microvascular thrombosis. It is unclear why [52].
Recently, another complement disorder has been described, the CHAPLE syndrome: Complement Hyperactivation Angiopathic thrombosis and Protein Losing Enteropathy. Loss of function mutations in CD55 causes a protein losing enteropathy presenting with abdominal pain, diarrhea and malabsorption. Small bowel biopsies show intestinal lymphangiectasia. 3 of 11 patients described had major vessel thrombosis including thrombosis in the IVC, right atrium, pulmonary artery, mesenteric vessels, hepatic and cerebral veins [61].
The wide spectrum of disorders of complement affecting both microvascular and macrovascular circulations, the complex interactions with the inflammatory and coagulation systems, points to the importance of the complement system in physiologic homeostasis and to the need for additional research to better understand these interactions [9,10,62-64].
Clinical course patient 3
Patient previously treated with cladribine chemotherapy for LGL leukemia who presented 1 week following bilateral Streptococcus Pneumonia with acute, severe, renal failure with an active urine sediment. The situation was further complicated by a positive urine drug screen for cocaine. The initial platelet count was normal. There was a moderate elevation of LDH. Bilirubin was normal. Initially only a rare schistocyte on smear. Initial considerations included post streptococcal glomerulonephritis, drug related acute kidney injury from recent antibiotics or cocaine, or Strep infection related HUS. A dialysis catheter was placed and dialysis initiated. ANCA serology for vasculitis was negative. HIV and acute hepatitis panels negative. By hospital day 6, platelets fell to 48,000, LDH rose to 706, indirect bilirubin up to 1.5 and haptoglobin down to 24. Blood film review now with increased schistocytes 1/HPF and the presence of spherocytes, consistent with a microangiopathic process. ADAM-TS enzyme returned at 44% of control. Diagnosis of HUS was established. Triple vaccination was given. Oral Cipro was started. Eculizumab weekly x 5 the Q 2 weeks started. Platelet count normalized by day 7 and LDH normalized within 2 weeks. Creatinine stabilized down to 2.4 at 5 months. Dialysis schedule decreased to twice weekly.
This case demonstrates the difficulty in establishing a diagnosis of aHUS from secondary HUS when multiple risk factors coexist. aHUS could have been triggered by the recent Streptococcal pneumonia which is felt to be both a primary and secondary precipitating factor for non Shiga toxin related secondary aHUS resulting from exposure of surface antigens on red cells and platelets subjecting them to complement mediated damage. Many drugs have been implicated as secondary causes, in this case, cocaine. In addition, this patient had an underlying lymphoproliferative disorder, LGL leukemia, which, although in remission, may have been a pre-disposing factor due to a compromised immune system.
Of note, the initial platelet count was normal and only a rare schistocyte was observed on the peripheral blood film. The disease process evolved over a number of days with a subsequent moderate fall in the platelet count, the appearance of numerous schistocytes in the peripheral blood, and increasing signs of hemolysis with a rise in serum LDH and fall in haptoglobin.
Conclusion
The thrombotic micoangiopathies are a heterogeneous group of blood disorders that typically present with a COOMB’s negative schistocytic hemolytic anemia with associated thrombocytopenia. Microvascular thrombi can cause severe, multifocal, end organ damage. The CNS, renal, GI, and cardiac microcirculations are frequent target organs. Signs and symptoms of disease may evolve over weeks to months and may not be present simultaneously at presentation. LDH elevation due to microvascular ischemia is frequently disproportionate to elevation of bilirubin and reticulocyte count. The level of ADAM-TS, Von Willebrand Factor cleaving metalloproteinase enzyme, is a key discriminator between TTP and HUS. Plasma exchange with or without steroids is the mainstay of treatment for TTP. Antibody therapies including Rituximab and Caplacizumab have the potential to further improve treatment outcomes. Integration of novel therapies into the frontline treatment of TTP is a work in progress.
Plasma exchange has no role in the treatment of Shiga toxin related HUS and only a limited role in the treatment of aHUS. Anti C5a complement antibody, eculizumab, is the preferred treatment of most subtypes of aHUS and has been successful in reversing or limiting chronic renal damage.
The Complement system is a complex system of proteins important in providing innate immunity which includes an “Alternate Pathway” of pathogen recognition independent of the antibody-antigen recognition system.
The Complement system has widespread interactions with other components of the acute inflammatory response system and the coagulation system which are currently being elucidated and may lead to the development of additional novel therapies.
Acknowledgements
The author would like to acknowledge all the scientists and clinicians who have contributed to this field, in particular Dr James George, Joel Moake, and Jeffrey Laurence, my clinical mentors who have always given graciously of their time and advice.
Conflict of Interest
The authors have no conflict of interests to disclose.
REFERENCES
- George JN, Nester CM. Syndromes of thrombotic microangiopathy. N Engl J Med. 2014;371:654-666.
- Moake JL. Studies on the pathophysiology of thrombotic thrombocytopenic purpura. Semin Hematol. 1997;34:83-89.
- Moake JL. Von Willebrand factor, ADAMTS-13, and thrombotic thrombocytopenic purpura. Semin Hematol. 2004;41:4-14.
- Veyradier A, Obert B, Houllier A, Meyer D, Girma JP. Specific von willebrand factor-cleaving protease in thrombotic microangiopathies: a study of 111 cases. Blood. 2001;98:1765-1772.
- Hovinga JAK, Lämmle B. Role of ADAMTS13 in the pathogenesis, diagnosis, and treatment of thrombotic thrombocytopenic purpura. ASH Educ Program Book 2012. 2012;610-616.
- Vesely SK, George JN, Lämmle B, Studt JD, Alberio L, El-Harake MA, et al. ADAMTS13 activity in thrombotic thrombocytopenic purpura-hemolytic uremic syndrome: relation to presenting features and clinical outcomes in a prospective cohort of 142 patients. Blood. 2003;102:60-68.
- Caprioli J, Noris M, Brioschi S, Pianetti G, Castelletti F, Bettinaglio P, et al. Genetics of HUS: the impact of MCP, CFH, and IF mutations on clinical presentation, response to treatment, and outcome. Blood. 2006;15:1267-1279.
- Noris M, Caprioli J, Bresin E, Mossali C, Pianetti G, Gamba S, et al. Relative role of genetic complement abnormalities in sporadic and familial aHUS and their impact on clinical phenotype. Clin J Am Soc Nephrol. 2010;5:1844-1859.
- Afshar-Kharghan V. Atypical hemolytic uremic syndrome. Hematol Am Soc Hematol Educ Program 2016. 2016;217-225.
- Jokiranta TS. HUS and atypical HUS. Blood. 2017;129:2847-2856.
- Risitano AM. Therapeutic complement modulation for hematological diseases: Where we stand and where we are going. Semin Hematol. 2018;55:113-117.
- Taylor RP, Lindorfer MA. Mechanisms of Complement-Mediated Damage in Hematological Disorders. Semin Hematol. 2018;55:118-123.
- George JN. How I treat patients with thrombotic thrombocytopenic purpura-hemolytic uremic syndrome. Blood 2000;96:1223-1229.
- Laurence J.Atypical hemolytic uremic syndrome (aHUS): Making the diagnosis. Clin Adv Hematol Oncol. 2012;10:1-12.
- Joly BS, Coppo P, Veyradier A. Thrombotic thrombocytopenic purpura. Blood. 2017;129:2836-2846.
- Scully M, Hunt BJ, Benjamin S, Liesner R, Rose P, Peyvandi F, et al. Guidelines on the diagnosis and management of thrombotic thrombocytopenic purpura and other thrombotic microangiopathies. Br J Haematol. 2012;158:323-335
- Stone JH. HELLP syndrome: hemolysis, elevated liver enzymes, and low platelets. JAMA. 1998;280:559-562.
- Abildgaard U, Heimdal K. Pathogenesis of the syndrome of hemolysis, elevated liver enzymes, and low platelet count (HELLP): a review. Eur J Obstet Gynecol Reprod Biol. 2012;166:117-123.
- Masias C, Vasu S, Cataland SR. None of the above: Thrombotic microangiopathy beyond TTP and HUS. Blood. 2017;129:2857-2863.
- Al-Nouri ZL, Reese JA, Terrell DR, Vesely SK, George JN. Druginduced thrombotic microangiopathy: a systematic review of published reports. Blood. 2015;125:616-618.
- Eremina V, Jefferson JA, Kowalewska J, Hochster H, Haas M, Weisstuch J, Richardson C, et al. VEGF inhibition and renal thrombotic microangiopathy. N Engl J Med. 2008;358:1129-1136.
- Kavanagh D, McGlasson S, Jury A, Williams J, Scolding N, Bellamy C, et al. Type I interferon causes thrombotic microangiopathy by a dosedependent toxic effect on the microvasculature. Blood 2016;128:2824-2833.
- Chapin J, Shore T, Forsberg P, Desman G, Van Besien K, Laurence J. Hematopoietic transplant-associated thrombotic microangiopathy: case report and review of diagnosis and treatments. Clin Adv Hematol Oncol. 2014;12:565-573.
- Hunt R, Yalamanoglu A, Tumlin J, Schiller T, Baek JH, Wu A, et al. A mechanistic investigation of thrombotic microangiopathy associated with IV abuse of Opana ER. Blood. 2017;129:896-905.
- George JN, Morton JM, Liles NW, Nester CM. After the party’s over. N Engl J Med. 2017;376:74-80.
- Grangé S, Bekri S, Artaud-Macari E, Francois A, Girault C, Poitou AL, et al. Adult-onset renal thrombotic microangiopathy and pulmonary arterial hypertension in cobalamin C deficiency. Lancet Lond Engl. 2015;386:1011-1012.
- Cornec-Le Gall E, Delmas Y, De Parscau L, Doucet L, Ogier H, Benoist JF, et al. Adult-onset eculizumab-resistant hemolytic uremic syndrome associated with cobalamin C deficiency. Am J Kidney Dis. 2014;63:119-123.
- Quaggin SE. DGKE and atypical HUS. Nat Genet. 2013;45:475-476.
- Bruneau S, Néel M, Roumenina LT, Frimat M, Laurent L, Frémeaux-Bacchi V, et al. Loss of DGKe induces endothelial cell activation and death independently of complement activation. Blood. 2015;125:1038-1046.
- Vijayan KV. DGKE disruption ditches complement and drives p38 signaling. Blood. 2015;125:898-899.
- Moake JL.Thrombotic microangiopathies. N Engl J Med. 2002;347:589-600.
- Laurence J, Haller H, Mannucci PM, Nangaku M, Praga M, Rodriguez de Cordoba S. Atypical hemolytic uremic syndrome (aHUS): essential aspects of an accurate diagnosis. Clin Adv Hematol Oncol. 2016;11:2-15.
- Jestin M, Benhamou Y, Schelpe AS, Roose E, Provôt F, Galicier L, et al. Preemptive rituximab prevents long-term relapses in immune-mediated thrombotic thrombocytopenic purpura. Blood. 2018;132:2143-2153.
- George JN. Clinical practice. Thrombotic thrombocytopenic purpura. N Engl J Med. 2006;354:1927-1935.
- Beloncle F, Buffet M, Coindre JP, Munoz-Bongrand N, Malot S, Pène F, et al. Splenectomy and/or cyclophosphamide as salvage therapies in thrombotic thrombocytopenic purpura: the French TMA Reference Center experience. Transfusion. 2012;52:2436-2444.
- George JN, Al-Nouri ZL. Diagnostic and therapeutic challenges in the thrombotic thrombocytopenic purpura and hemolytic uremic syndromes. Hematol Am Soc Hematol Educ Program 2012;604-609.
- Sarode R, Bandarenko N, Brecher ME, Kiss JE, Marques MB, Szczepiorkowski ZM. Thrombotic thrombocytopenic purpura: 2012 American Society for Apheresis (ASFA) consensus conference on classification, diagnosis, management, and future research. J Clin Apher. 2014;29:148-167.
- Sayani FA, Abrams CS. How I treat refractory thrombotic thrombocytopenic purpura. Blood 2015;125:3860-3867.
- Lim W, Vesely SK, George JN. The role of rituximab in the management of patients with acquired thrombotic thrombocytopenic purpura. Blood 2015;125:1526-1531.
- Blitzer JB, Granfortuna JM, Gottlieb AJ, Smith JR, Theodorakis ME, Zamkoff KW, et al. Thrombotic thrombocytopenic purpura: treatment with plasmapheresis. Am J Hematol. 1987;24:329-339.
- Zheng X, Pallera AM, Goodnough LT, Sadler JE, Blinder MA. Remission of chronic thrombotic thrombocytopenic purpura after treatment with cyclophosphamide and rituximab. Ann Intern Med. 2003;138:105-108.
- Westwood JP, Webster H, McGuckin S, McDonald V, Machin SJ, Scully M. Rituximab for thrombotic thrombocytopenic purpura: benefit of early administration during acute episodes and use of prophylaxis to prevent relapse. J Thromb Haemost. 2013;11:481-490.
- Elliott MA, Heit JA, Pruthi RK, Gastineau DA, Winters JL, Hook CC. Rituximab for refractory and or relapsing thrombotic thrombocytopenic purpura related to immune-mediated severe ADAMTS13-deficiency: a report of four cases and a systematic review of the literature. Eur J Haematol. 2009;83:365-372.
- Froissart A, Buffet M, Veyradier A, Poullin P, Provot F, Malot S, et al. Firstline rituximab efficacy and safety in patients with acquired idiopathic thrombotic thrombocytopenic purpura experiencing a non optimal response to therapeutical plasma exchange: Results of a prospective multicenter phase 2 study from the french reference center for the management of thrombotic microangiopathies. Blood. 2009;114:890.
- Bhagirath VC, Kelton JG, Moore J, Arnold DM. Rituximab maintenance for relapsed refractory thrombotic thrombocytopenic purpura. Transfusion. 2012;52:2517-2523.
- Kappers-Klunne MC, Wijermans P, Fijnheer R, Croockewit AJ, van der Holt B, de Wolf JT, et al. Splenectomy for the treatment of thrombotic thrombocytopenic purpura. Br J Haematol. 2005;130:768-776.
- Aqui NA, Stein SH, Konkle BA, Abrams CS, Strobl FJ. Role of splenectomy in patients with refractory or relapsed thrombotic thrombocytopenic purpura. J Clin Apheresis 2003;18:51-54.
- Schwartz J, Eldor A, Szold A (2001) Laparoscopic splenectomy in patients with refractory or relapsing thrombotic thrombocytopenic purpura. Arch Surg Chic Ill. 2001;136:1236-1238.
- Scully M, Knöbl P, Kentouche K, Rice L, Windyga J, Schneppenheim R, et al. Recombinant ADAMTS-13: first-in-human pharmacokinetics and safety in congenital thrombotic thrombocytopenic purpura. Blood. 2017;130:2055-2063.
- Veyradier A. Von willebrand factor-A new target for TTP treatment? N Engl J Med. 2016;374:583-585.
- Peyvandi F, Scully M, Kremer Hovinga JA, Cataland S, Knöbl P, Wu H, et al.Caplacizumab for acquired thrombotic thrombocytopenic purpura. N Engl J Med. 2016;374:511-522.
- Scully M, Cataland SR, Peyvandi F, Coppo P, Knöbl P, Kremer Hovinga JA, et al. Caplacizumab treatment for acquired thrombotic thrombocytopenic turpura. N Engl J Med 2019;380:335-346.
- Chen J1, Reheman A, Gushiken FC, Nolasco L, Fu X, Moake JL, et al. N-acetylcysteine reduces the size and activity of von Willebrand factor in human plasma and mice. J Clin Invest. 2011;121:593-603.
- Li GW, Rambally S, Kamboj J, Reilly S, Moake JL, Udden MM, et al. Treatment of refractory thrombotic thrombocytopenic purpura with Nacetylcysteine: A case report. Transfusion. 2014;54:1221-1224.
- Shortt J, Opat SS, Wood EM. N-Acetylcysteine for thrombotic thrombocytopenic purpura: is a von Willebrand factor-inhibitory dose feasible in vivo? Transfusion. 2014;54:2362-2363.
- Shortt J, Oh DH, Opat SS. ADAMTS13 antibody depletion by bortezomib in thrombotic thrombocytopenic purpura. N Engl J Med. 2013;368:90-92.
- Sadler JE. Pathophysiology of thrombotic thrombocytopenic purpura. Blood. 2017;130:1181-1188.
- Plaimauer B, Kremer Hovinga JA, Juno C, Wolfsegger MJ, Skalicky S, Schmidt M, et al. Recombinant ADAMTS13 normalizes von Willebrand factor-cleaving activity in plasma of acquired TTP patients by overriding inhibitory antibodies. J Thromb Haemost JTH. 2011;9:936-44.
- Khanal N, Dahal S, Upadhyay S, Bhatt VR, Bierman PJ. Differentiating malignant hypertension-induced thrombotic microangiopathy from thrombotic thrombocytopenic purpura. Ther Adv Hematol. 2015;6:97-102.
- Shibagaki Y, Fujita T. Thrombotic microangiopathy in malignant hypertension and hemolytic uremic syndrome (HUS)/ thrombotic thrombocytopenic purpura (TTP): can we differentiate one from the other? Hypertens Res Off J Jpn Soc Hypertens. 2005;28:89-95.
- Timmermans SAMEG, Abdul-Hamid MA, Vanderlocht J. Patients with hypertension-associated thrombotic microangiopathy may present with complement abnormalities. Kidney Int 2017;91:1420-1425.
- Fakhouri F, Loirat C. Anticomplement treatment in atypical and typical hemolytic uremic syndrome. Semin Hematol. 2018;55:150-158.
- Fakhouri F, Zuber J, Frémeaux-Bacchi V, Loirat C. Haemolytic uraemic syndromeLancet Lond Engl. 2017;390:681-696.
- Laurence J. Atypical hemolytic uremic syndrome (aHUS): Treating the patient. Clin Adv Hematol Oncol HO. 2013;11:3-15.
- Nester CM, Thomas CP. Atypical hemolytic uremic syndrome: what is it, how is it diagnosed, and how is it treated? Hematol Am Soc Hematol Educ Program 2012. 2012;617-625.
- Moake JL. A new therapeutic strategy for atypical HUS. Blood. 2017;130:243-244.
- Grabowski EF. The hemolytic-uremic syndrome-toxin, thrombin, and thrombosis. N Engl J Med. 2002;346:58-61.
- Konar M, Granoff DM. Eculizumab treatment and impaired opsonophagocytic killing of meningococci by whole blood from immunized adults. Blood. 2017;130:891-899.
- Thielen AJF, Zeerleder S, Wouters D. Consequences of dysregulated complement regulators on red blood cells. Blood Rev. 2018;32:280-288.
- Nishimura J, Yamamoto M, Hayashi S. Genetic variants in C5 and poor response to eculizumab. N Engl J Med. 2014;370:632-639.
- Antoine G, Zimmermann K, Plaimauer B. ADAMTS13 gene defects in two brothers with constitutional thrombotic thrombocytopenic purpura and normalization of von Willebrand factor-cleaving protease activity by recombinant human ADAMTS13. Br J Haematol. 2013;120:821-824.
- Salmon JE, Heuser C, Triebwasser M. Mutations in complement regulatory proteins predispose to preeclampsia: a genetic analysis of the PROMISSE cohort. PLoS Med. 2011;8:e1001013.
- Burwick RM, Feinberg BB. Eculizumab for the treatment of preeclampsia/HELLP syndrome. Placenta. 2012;34:201-203.
- Sicre de FF, Peffault de LR. Ten years of clinical experience with eculizumab in patients with paroxysmal nocturnal hemoglobinuria. Semin Hematol. 2018;55:124-129.
- Legendre CM, Licht C, Muus P.Terminal complement inhibitor eculizumab in atypical hemolytic-uremic syndrome. N Engl J Med. 2013;368:2169-2181.
- https://www.accessdata.fda.gov/drugsatfda_docs/label/2007/125166lbl.pdf
- Loirat C, Fakhouri F, Ariceta G. An international consensus approach to the management of atypical hemolytic uremic syndrome in children. Pediatr Nephrol Berl Ger. 2016;31:15-39.
- Merrill SA, Brittingham ZD, Yuan X, Moliterno AR, Sperati CJ, Brodsky RA. Eculizumab cessation in atypical hemolytic uremic syndrome. Blood. 2017;130:368-372.
- Macia M, De Alvaro MF, Dutt T. Current evidence on the discontinuation of eculizumab in patients with atypical haemolytic uraemic syndrome. Clin Kidney J. 2017;10:310-319.
- Nürnberger J, Philipp T, Witzke O. Eculizumab for atypical hemolytic-uremic syndrome. N Engl J Med. 2009;360:542-544.
- Gruppo RA, Rother RP. Eculizumab for congenital atypical hemolytic-uremic syndrome. N Engl J Med. 2009;360:544-546.
- Noris M, Remuzzi G. Atypical Hemolytic–Uremic Syndrome. N Engl J Med. 2009;361:1676.
Citation: Granfortuna J (2019) The Many Faces of Thrombotic Microangiopathies. J Blood Disord Transfus 10:422.
Copyright: © 2019 Granfortuna J. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

