Indexed In
- Euro Pub
- Google Scholar
Useful Links
Share This Page
Journal Flyer

Open Access Journals
- Agri and Aquaculture
- Biochemistry
- Bioinformatics & Systems Biology
- Business & Management
- Chemistry
- Clinical Sciences
- Engineering
- Food & Nutrition
- General Science
- Genetics & Molecular Biology
- Immunology & Microbiology
- Medical Sciences
- Neuroscience & Psychology
- Nursing & Health Care
- Pharmaceutical Sciences
Research Article - (2025) Volume 9, Issue 1
Influence of follicular fluid metals on female sexual hormones
Jorge Gómez-Rodríguez2, Rubí Rodríguez-Díaz1,2*, Raquel Blanes-Zamora1,2, Soraya Paz-Montelongo3, Samuel Alejando Vega3, Jonay González-Pérez1,2, Ángel J. Gutiérrez3, Carmen Rubio3, Arturo Hardisson3 and Enrique González-Dávila42Department of Obstetrics and Gynecology, Universidad de La Laguna, Tenerife, Spain
3Department of Toxicology, Universidad de La Laguna, Tenerife, Spain
4Department of Mathematics, Statistics and Operations Research, IMAULL, Universidad de La Laguna, Tenerife, Spain
Received: 28-Feb-2025, Manuscript No. JCMS-25-28483; Editor assigned: 03-Mar-2025, Pre QC No. JCMS-25-28483 (PQ); Reviewed: 17-Mar-2025, QC No. JCMS-25-28483; Revised: 24-Feb-2025, Manuscript No. JCMS-25-28483 (R); Published: 31-Mar-2025, DOI: 10.35248/2593-9947.25.9.304
Abstract
There are chemicals, such as metals, that act as disruptors in the ovarian follicle. This study examines the impact of microelement, macroelement and heavy metals, in Follicular Fluid (FF) on female sex hormones in women undergoing assisted reproduction treatments in the Canary Islands. The research aims to better understand the relationship between metals and female reproductive function, focusing on how these elements impact hormone levels. The content of twenty metals (Al, B, Ba, Ca, Cd, Co, Cr, Cu, K, Fe, Li, Mg, Mn, Mo, Na, Ni, Pb, Sr, V and Zn) were measured in the FF of 63 participants using ICP-OES (inductively coupled plasma optical emission spectrometry). Serum levels of AMH, FSH, LH and estradiol (E2) were determined to assess hormone levels. A direct relationship of FSH with Mn (p=0.040) and close to significance with Fe (p=0.081) was obtained. LH was related to Sr (p=0.020) and with Mg (p=0.042) and was close to significance with K (p=0.083) and Na (p=0.086). An inverse relationship was identified between E2 and K (p=0.003), Ca (p=0.023) and Na (p=0.016). Fe levels were significantly lower in the low AMH group (≤ 1.1 ng/mL) (p<0.044). Ca (p<0.002) and Na (p<0.002) were significantly higher in the low AMH group, while K showed a non-significant trend. Low AMH is associated to older age and a trend toward higher BMI. Understanding metal effects on hormones could have significant implications for reproductive health.
Keywords
Follicular fluid; Microelement metals; Heavy metals; Macroelement metals; AMH; Female sexual hormones
Introduction
The presence of metals may have a marked effect on ovarian reserve and may also affect sex hormone levels [1] .There are metals, such as Heavy Metals (HM) or toxic metals, including Lead (Pb), Mercury (Hg), Aluminium (Al) and Cadmium (Cd), that act as disruptors of the ovarian follicle, with detrimental outcomes on endocrine capacity and the viability of theca and granulosa cells [2]. The action mechanisms of HM are not clear, inflammation and oxidative stress have been proposed as the main possible aetiologies, also affect homeostasis, resulting in endocrine imbalance, and leading to ovarian follicular atresia [3-6].
Although several microelement metals, such as Zinc (Zn), Nickel (Ni), Iron (Fe), Cobalt (Co), Copper (Cu), Molybdenum (Mo), Manganese (Mn), and Chromium (Cr), are essential for human health, they can be toxic at high concentrations, however, HMs are always toxic [7].
Exposure to HM, macroelement, such as Calcium (Ca), Potassium (K), Magnesium (Mg) and Sodium (Na), and microelements, occurs through food, water, air, waste, and cosmetics, with dietary intake being the main source of exposure [8,9].
Environmental factors, with HM being a possible causative agent, are one of the etiological causes of Diminished Ovarian Reserve (DOR), which is characterized by the progressive loss of ovarian primordial follicles, and a decline of Anti-Müllerian Hormone (AMH) [10-12]. Its prevalence has increased over the last decade [13]. It is thought that AMH levels <1.1 ng/ml, in addition to antral follicle counts <5-7, are associated with DOR, which also includes high Follicle-Stimulating Hormone (FSH) levels, a high FSH/Luteinizing Hormone (LH) ratio, and a low number of oocytes retrieved during Assisted Reproductive Treatment (ART) [10-15].
Lee, et al. observed that the exposure to metals may alter AMH levels in premenopausal women and act as an endocrine disruptor [16]. In addition, the influence of residence has also been identified, as the bioaccumulation of Fe and Co was significantly different between infertile women living in eastern compared to southern China, who were reported to have significantly higher Fe and Co Follicular Fluid (FF) levels and a lower AMH levels [17]. Several authors identified nine articles that investigated the relationship between HMs and those female hormone levels, and a recent publication examined macroelements such as Ca, K, and Na, and the essential microelement Fe, in relation to AMH [18,19].
In order to study the effect of metals on the ovary, many authors analyse its presence in blood or even in urine but considering the Blood-Follicular Barrier (BFB), the determination in FF is much more accurate [20]. The FF consists of an ultrafiltration of blood plasma and may contain essential and non-essential metals, but high molecular weight proteins do not cross the BFB. This barrier is selective for both molecular size and ionic charge and hormones can also affect this selective permeability [21].
The FF environment can best indicate exposure to the real microenvironment surrounding the growing follicle and that can best be correlated with the follicular activity. Therefore, the present study was designed with the determination of metals in FF as the preferred option. Some authors have shown that the avoidance of certain HM and the appropriate supplementation of essential microelements are beneficial for ovarian function [19].
Therefore, the main objective of this research is to better understand the relationship between metals and female reproductive function, focusing on how these elements affect hormone levels and ovarian follicle activity. This study will also analyse the relationship between metals and sex hormones in females in the Canary Islands.
Materials and Methods
Samples
A cross-sectional study was carried out on 63 females who underwent ART, in the Reproduction Unit of the Hospital Universitario de Canarias, between February 2019 and December 2020. The 63 female FF samples correspond to the number of patients who attended successively for ART, during this period. Twenty metals were measured in the FF. These patients also underwent a prospective study evaluating the results of ART.
The authors declare that all procedures contributing to this work complied with the ethical standards of the relevant national and institutional human experimentation committees and with the Helsinki Declaration of 1975, as revised in 2008. The study protocol was reviewed and approved by the Institutional Review Board of the Hospital Universitario de Canarias (Reg. No. CHUC 2018 53), and all eligible participants signed an informed consent form prior to enrolment.
Sample treatment
Hormone levels determination: Sample selection and clinical protocols were as previously described [22]. Tobacco users were categorized as never smokers, and smokers [23]. Alcohol consumers were categorized into no consumption, and consumption [24]. And the WHO classification was followed for BMI: Normal range 18.5- 24.9, overweight/ obese 25- ≥ 30.0 [25].
Hormone levels were measured using commercially available kits. The AMH assay was based on an immunoassay (Roche Elecsys® AMH Assay). The range of serum AMH levels, at any phase of the cycle and based on the Bologna criteria, was defined as low AMH (19%) when AMH ≤ 1.1 ng/mL, and normal (80.9%) when AMH>1.1 ng/mL [4]. In order to study the influence of metals on AMH, we divided the participants into two groups according to these criteria. FSH, LH, and estradiol (E2) levels were determined in the early follicular phase (day 3-5), by a chemiluminescence technique (Immulite 2000®, Siemens).
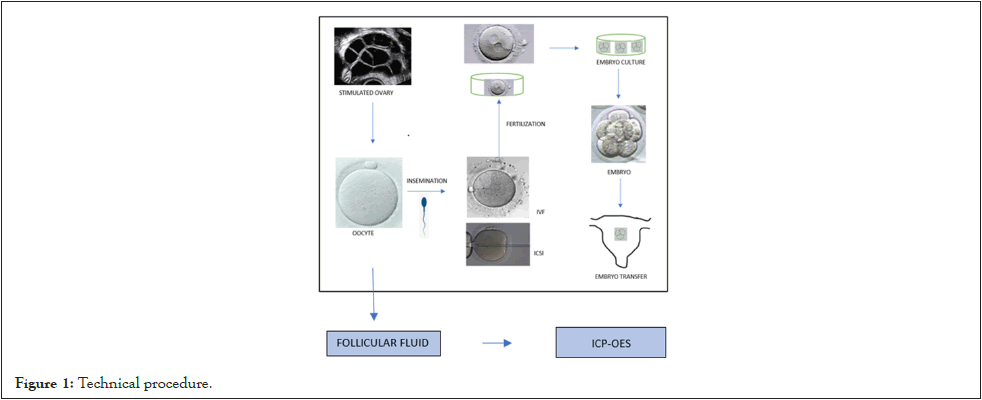
Participants underwent controlled ovarian stimulation, and FF aspiration was conducted by a transvaginal puncture, guided by transvaginal ultrasound. The oocytes were then inseminated by In Vitro Fertilization (IVF) or Intracytoplasmic Sperm Injection (ICSI), and the FF was stored in a refrigerator until processed for metal detection. After oocyte insemination, oocyte fertilisation and the embryo cleavage follow up were accomplished. According to the ASEBIR criteria, a good prognosis is considered when the Fertilization Rate (FR) is >75%, the Cleavage Rate (CR) is 100%, and the Blastocyst Rate (BR) is >50%. The embryo Implantation Rate (IR) is defined as the percentage of embryos transferred to the uterus that resulted in pregnancy, and the Pregnancy Rate (PR) is defined as the number of cycles with at least one gestational sac vs. the number of cycles with Embryo Transfer (ET) (Figure 1) [26].
Figure 1: Technical procedure.
Metals determination: Metal detection was carried out in the Canary Islands Health Service in collaboration with the Area of Toxicology of the University of La Laguna.
A total of 63 samples were analysed using an Inductively Coupled Plasma Optical Emission Spectrometer (ICP-OES), model ICPOES Thermo Scientific iCAP PRO (Waltham, MA, USA) with an automated autosampler (Auto Sampler, CETAX model ASX- 520). The samples were previously digested using a microwave digester (Multiwave GO, Anton Paar GO, Austria). The technical procedure has been described previously [22].
The following metals were measured in the FF: Al, Boron (B), Barium (Ba), Ca, Cd, Co, Cr, Cu, Fe, K, Lithium (Li), Mg, Mn, Mo, Na, Ni, Pb, Strontium (Sr), Vanadium (V) and Zn. The instrumental limits of detection and quantification were estimated from the instrumental response, specifically, they were determined by analysing fifteen blanks under reproducible conditions (Table 1).
| Metal | Wavelength (nm) | LOD (mg/L) | LOQ (mg/L) |
|---|---|---|---|
| Al | 167.0 | 0.005 | 0.015 |
| B | 249.6 | 0.008 | 0.027 |
| Ba | 455.4 | 0.0006 | 0.002 |
| Ca | 315.8 | 1.629 | 5.432 |
| Cd | 214.4 | 0.0007 | 0.002 |
| Co | 228.6 | 0.001 | 0.005 |
| Cr | 267.7 | 0.001 | 0.005 |
| Cu | 324.7 | 0.003 | 0.011 |
| K | 766.4 | 1.764 | 5.883 |
| Fe | 259.9 | 0.003 | 0.009 |
| Li | 670.7 | 0.013 | 0.031 |
| Mg | 383.8 | 1.58 | 5.268 |
| Mn | 257.6 | 0.0008 | 0.003 |
| Mo | 202.0 | 0.0016 | 0.005 |
| Na | 818.3 | 2.221 | 7.404 |
| Ni | 221.6 | 0.0009 | 0.003 |
| Pb | 220.3 | 0.0009 | 0.003 |
| Sr | 407.7 | 0.003 | 0.011 |
| V | 292.4 | 0.0014 | 0.004 |
| Zn | 213.8 | 0.0027 | 0.009 |
Table 1: Limits of Detection (LOD) and Limit of Quantification (LOQ) for each metal level.
Standard solutions were prepared using 65% Merck nitric acid. For the alkaline and alkaline earth metals (Na, Ca, K, Mg), the certified IV-STOCK-2 standard from Inorganic Ventures (Inorganic Ventures Inc., Christiansburg, VA, USA) was used with a concentration of 10000 μg/ml. For the rest of the metals analyzed, the Multi-Element Std SCP28AES certified standard from SCP Science was used (SCP28AES, SCP Science, Quebec, Canada), with a concentration of 10 mg/l per element. These standards guarantee the precision and traceability of the analytical results.
As this was a descriptive study, the validation criteria were evaluated at the limit of quantification and on certified reference materials. The validation parameters have been defined as established in commission regulation (EC) No 333/2007 [27]. Specifically, the following have been evaluated:
• Specificity: It has been confirmed that the method does not present spectral interferences for the metals analyzed in follicular fluid.
• Precision: Evaluated as reproducibility, it has been checked for each metal, obtaining a HORRATR value lower than 2.
• Accuracy: Determined through recovery, values between 85% and 110% were obtained for all samples. The lowest recoveries were observed in samples spiked to the limit of quantification. It should be noted that it has not been necessary to apply correction or recovery factors, since by using reference materials it has been demonstrated that the certified reference concentration is reached, both in natural and spiked samples.
These parameters have been verified by measuring, under reproducibility conditions, 10 fortified FF samples at the limit of quantification for each metal, and by 10 repeated measurements of the reference materials, also used as quality controls of the method.
Data analysis
Data were summarized as relative frequencies for categorical variables, means ± standard deviation for normally distributed variables and medians (interquartile range IQR, P25; P75) for nonnormal data.
Depending on the type of variable and the number of groups to be compared, comparisons were made using Pearson's chi-squared test, the Kruskal-Wallis test or the Mann-Whitney U test and ANOVA or t-student.
The relationship between the continuous variables was calculated using Pearson’s or Spearman’s correlation coefficient. A power analysis was performed for the sample sizes used and the 95% confidence level, with the detection of a difference between the values of the metals equal to one standard deviation of the power being 81.1%. Metal content thresholds or Receiver Operating Characteristics (ROC) curve cut-off points were determined using the Youden index criterion.
Logistic regression with Wald's backward variable selection method was then used to model the probability of presenting AMH ≤ 1.1 ng/mL. The explanatory or independent variables that showed a p<0.1 in the univariate analysis were selected. The ROC curves and the Area under the Curve (AUROC) were considered.
The Akaike Information Criterion (AICc) value and the percentage of agreement were reported. SPSS V 29 (IBM SPSS Statistics) and MedCalc V 19.5 (MedCalc Software Ltd.) were used and a p-value ≤ 0.05 was considered significant.
Results
Demographic and clinical characteristics
Female participants ranged in age from 27 to 42 years, all residing in the Canary Islands, Spain, with a mean age of 36.3 ± 3.4 years and a BMI of 24.8 ± 3 kg/m2. Regarding the gynaecological history of the participants, 22% had Polycystic Ovaries (PCO), 15.5% had tubal pathology, 3.1% had fibroids, 3.1% had endometriosis, and 1.5% had cervical pathology. Of these participants, 66.7% had primary infertility, 4.7% had a secondary infertility, and 17.5% reported a previous pregnancy loss. All the participants followed the Mediterranean diet, and 46% were overweight or obese. All patients received supplements containing folic acid, vitamins B12 and D, iodine, and Zn (15 mg per day), as a nutritional supplement that helps meet a woman's nutritional needs from the moment she starts planning a pregnancy. Despite Zn supplementation, we did not find any remarkable results for this metal (Table 2).
| Characteristic | Total |
|---|---|
| Age (years) | 36.3 ± 3.4 |
| BMI (kg/m2) | 24.8 ± 3.4 |
| BMI, n (%) | |
| (a) Low/normal | 33 (53) |
| (b) Overweight/Obese | 29 (47) |
| Tobacco, n (%) | 9 (14) |
| Alcohol, n (%) | 20 (32) |
| FSH mUI/mL | 6.7 ± 1.9 |
| LH mUI/mL | 5.3±2.5 |
| E2 pg/mL | 55.8 ± 25.2 |
Table 2: Female characteristics.
Twelve of the participants (19%) had AMH levels ≤ 1.1 ng/mL, 29 (46%) had levels between 1.1 and 3 ng/mL, and 22 (35%) had levels above 3 ng/mL. The values ranged from 0.49 to 7.94 ng/ mL, and the mean serum AMH was 2.5 ± 1.7 ng/mL, the median was 1.8 ng/mL (IQR was 1.2, 3.5). The mean age in the low AMH group was 38.3 ± 1.6, for AMH between 1.1-3 ng/mL the main age was 36.2 ± 3.6 years, and when AMH was >3 ng/mL, the mean age was 35.3 ± 3.6 years. The age difference between low and normal AMH was statistically significant, 38.3 ± 1.6 vs. 35.8 ± 3.6, 1.07 (95% CI 0.40, 4.66) years older in low AMH participants (p<0.021). AMH was higher in normal weight participants, and in the low AMH group, only 27% were normal weight, 73% were overweight or obese compared to 41% of the normal AMH participants and showed a trend toward significance (p<0.057). No association was established between toxic habits, such as tobacco and alcohol (Table 3).
| Characteristic | AMH | P | |
|---|---|---|---|
| ≤ 1.1 | > 1.1 | ||
| Age (years) | 38.3 ± 1.6 | 35.8 ± 3.6 | 0.021 |
| BMI (kg/m2) | 26.2 ± 3.1 | 24.5 ± 3.4 | 0.117 |
| BMI, n (%) | 0.057 | ||
| Low/normal | 3 (27) | 30 (59) | |
| Overweight/Obese | 8 (73) | 21 (41) | |
| Tobacco, n (%) | 1 (8) | 8 (16) | 0.844 |
| Alcohol, n (%) | 5 (42) | 15 (29) | 0.496 |
| FSH mUI/mL | 6.9 ± 1.9 | 6.7 ± 1.9 | 0.78 |
| LH mUI/mL | 4.0 ± 1.4 | 5.7± 2.6 | 0.035 |
| E2 pg/mL | 55.7 ± 21.8 | 55.9 ± 26.4 | |
Note: *Median (interquartile range) (P25; P75)
Table 3: Female characteristics in relation to AMH level.
Metals
Almost all the participants presented Cu, Fe, Zn, Ca, K, and Na, and there were only two participants with Pb and Sr, while the remaining metals were identified in few participants. There were several metals: Co, Mo, Cr, B, Cd, Li and V, which were not observed in any of the patients (Table 4).
| Metal | Total |
|---|---|
| Heavy metals, n (%) | |
| Pb | 2 (2) |
| Cd | - |
| Al | 44 (70) |
| Al (mg/kg) | 0.70 ± 0.78 |
| Al (mg/kg)* | 0.50 (0.32; 0.80) |
| Microelement metals, n (%) | |
| Essential metals | |
| Co | - |
| Mo | - |
| Cr | - |
| Cu | 56 (89) |
| Fe | 59 (94) |
| Zn | 56 (89) |
| Mn | 40 (64) |
| Cu (mg/kg) | 0.90 ± 0.31 |
| Fe (mg/kg) | 14.86 ± 13.17 |
| Fe (mg/kg)* | 10.0 (6.7; 17.0) |
| Zn (mg/kg) | 1.14 ± 0.37 |
| Mn (mg/kg) | 0.04 ± 0.01 |
| Non essential metals | |
| Ba | 7 (11) |
| B | - |
| Li | - |
| Ni | 5 (8) |
| Sr | 2 (2) |
| V | - |
| Macroelement metals, n (%) | |
| Ca | 57 (90) |
| K | 57 (90) |
| Mg | 8 (13) |
| Na | 61 (97) |
| Ca (mg/kg) | 136.2 ± 66.9 |
| Ca (mg/kg) * | 147 (93; 180) |
| K (mg/kg) | 300.9 ± 149.6 |
| K (mg/kg) * | 305 (233; 352) |
| Mg (mg/kg) | 67.9 ± 5.9 |
| Na (mg/kg) | 4475 ± 1617 |
| Na (mg/kg)* | 4869 (3286; 5194) |
Note: *Median (interquartile range)
Table 4: Content and presence of metals (mg/Kg).
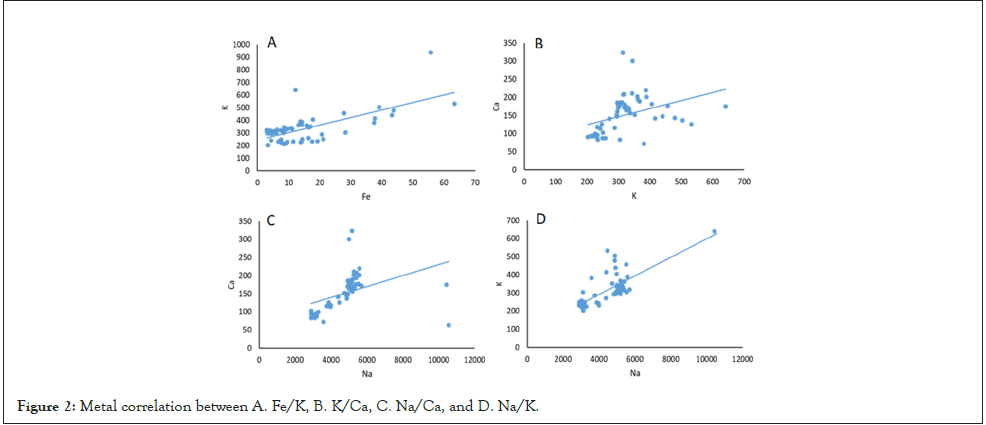
A correlation was found between some metals, such as Fe and K (rs=0.65, p<0.001); Ca and K (rs=0.50, p<0.001); Ca and Na (rs=0.53, p<0.001); and K and Na (r2=0.784, p<0.001) (Figure 2).
Figure 2: Metal correlation between A. Fe/K, B. K/Ca, C. Na/Ca, and D. Na/K.
Fe levels were significantly lower in the low AMH group, and Ca and Na levels were significantly higher, with K levels also showing a tendency to be higher. Half of the normal AMH participants had Fe levels >11.5 mg/kg, compared to 5.7 mg/kg in the low AMH participants (p<0.044). Ca was found to be 125 vs. 179 mg/kg in the normal AMH group vs. the low AMH group (p<0.002). The concentration of K was 297 vs. 316 mg/kg (p<0.093), and that of Na was 4374 vs. 5226 mg/kg (p<0.002). The presence of Ca was 125 mg/kg (91; 175, p<0.002) and that of Na was 4374 mg/kg (3132; 5061, p<0.002), with this having a higher statistical significance in the low AMH group, which had the worst reproductive results (Table 5).
| Metal | AMH | P | |
|---|---|---|---|
| ≤ 1.1 (N=12) |
> 1.1 (N=51) |
||
| Heavy metals, n (%) | |||
| Pb | - | 1 (2) | 1 |
| Cd | - | - | |
| Al | 9 (75) | 35 (69) | 0.934 |
| Al (mg/kg) | 0.47 ± 0.18 | 0.76 ± 0.86 | 0.316 |
| Al (mg/kg) * | 0.41 (0.34; 0.62) | 0.53 (0.32; 0.90) | 0.314 |
| Microelement metals, n (%) | |||
| Essential metals, n (%) | |||
| Co | - | - | |
| Mo | - | - | |
| Cr | - | - | |
| Cu | 12 (100) | 44 (86) | 0.329 |
| Fe | 12 (100) | 47 (92) | 0.73 |
| Zn | 12 (100) | 44 (86) | 0.329 |
| Mn | 8 (67) | 32 (63) | 0.8 |
| Cu (mg/kg) | 0.87 ± 0.24 | 0.91 ± 0.33 | 0.723 |
| Fe (mg/kg) | 13.77 ± 18.49 | 15.16 ± 11.68 | 0.735 |
| Fe (mg/kg)* | 5.7 (4.1; 12.5) | 11.5 (7.8; 17.6) | 0.044 |
| Zn (mg/kg) | 1.14 ± 0.26 | 1.14 ± 0.40 | 0.991 |
| Mn (mg/kg) | 0.03 ± 0.01 | 0.04 ± 0.01 | 0.314 |
| Non essential metals, n (%) | |||
| Ba | 1 (8) | 6 (12) | 0.734 |
| B | - | - | |
| Li | - | - | |
| Ni | - | 5 (10) | 0.573 |
| Sr | - | 1 (2) | 1 |
| V | - | - | |
| Macroelement metals, n (%) | |||
| Ca | 12 (100) | 45 (88) | 0.585 |
| K | 12 (100) | 45 (88) | 0.585 |
| Mg | 1 (8) | 7 (14) | 0.614 |
| Na | 12 (100) | 49 (96) | 0.653 |
| Ca (mg/kg) | 188.3 ± 51.4 | 124.0 ± 64.6 | 0.002 |
| Ca (mg/kg)* | 179 (153; 209) | 125 (91; 175) | 0.002 |
| K (mg/kg) | 352.5 ± 82.1 | 288.7 ± 159.6 | 0.186 |
| K (mg/kg) * | 316 (299; 379) | 297 (231; 351) | 0.093 |
| Mg (mg/kg) | 64.6 | 68.4 ± 6.2 | 0.589 |
| Na (mg/kg) | 5203 ± 353 | 4304 ± 1749 | 0.083 |
| Na (mg/kg) * | 5226 (4939; 5499) | 4374 (3132; 5061) | 0.002 |
Note: *Median (interquartile range)
Table 5: Study of the content and presence of metals based on the AMH level.
For those metals that were compared (parametrically or nonparametrically), the values differed depending on whether AMH was chosen >1.1 ng/mL with a p-value<0.10. Fe, Ca, K, and Na were selected to evaluate the possibility of defining a cut-off point that would allow the discrimination between the two groups. The cut-off points were obtained according to Youden's index, and the age variable was also included (Table 6).
| CUT-OFF POINTS | AMH ng/mL | P | OR (IC95%) | AUROC | |
|---|---|---|---|---|---|
| ≤ 1.1 | > 1.1 | ||||
| Fe ≤ 6.1 mg/Kg | 7 (58) | 10 (20) | 0.012 | 5.7 (1.5; 21.9) | 0.64 (0.51; 0.75) |
| Ca>125.47 mg/Kg | 12 (100) | 25 (49) | 0.001 | 0.76 (0.67; 0.88) | |
| K>287.91 mg/kg | 12 (100) | 27 (53) | 0.002 | 0.66 (0.53; 0.77) | |
| Na>5116.04 mg/Kg | 9 (75) | 11 (22) | 0.001 | 10.9 (2.5; 47.3) | 0.80 (0.68; 0.89) |
| Age>35 | 12 (100) | 30 (59) | 0.006 | 0.71 (0.58; 0.82) | |
Table 6: Cut-off points (Youden´s index) favoring AMH ≤ 1.1 ng/mL.
This model gave an AUROC of 0.89 (CI95% 0.78; 0.95; p<0.001) (Table 7).
| B | s.e. | P | OR (95% CI) | |
|---|---|---|---|---|
| Fe ≤ 6.1 mg/kg | 2.147 | 0.906 | 0.018 | 8.555 (1.450; 50.484) |
| Na>5116.04 mg/kg | 2.373 | 0.862 | 0.006 | 10.729 (1.982; 58.095) |
| Age | 0.408 | 0.198 | 0.04 | 1.504 (1.019; 2.219) |
| Constant | -18.562 | 7.705 | 0.016 |
Note: OR: Odds Ratio; s.e: standard error; CI: Confidence Interval
Table 7: Logistic regression model to predict the probability of AMH ≤ 1.1 ng/mL.
The prognostic probability of AMH ≤ 1.1, was obtained as follows;
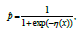 where the linear predictor is: Õ²(x)=-18.562+2.373 × (if Na>5116.04) + 2.147 × (if Fe ≤ 6.1)+0.408 × age. According to Youden's criterion, the test is considered positive if pˆ > 9.63%, with being the estimated probability of AMH ≤ 1.1 ng/mL, with a sensitivity of 92% and a specificity of 78%. If the prevalence of having an AMH below 1.1 ng/mL is the one found in the sample used, 19%, the positive predictive value would be 50% (95% CI 36.6; 63.4) and the negative predictive value would be 97.6% (95% CI 85.9; 99.6). The latter result would indicate that this model is better for ruling out AMH levels (if, pˆ > 9.63%,97.6% have AMH greater than 1.1 ng/mL) than for confirming them.
where the linear predictor is: Õ²(x)=-18.562+2.373 × (if Na>5116.04) + 2.147 × (if Fe ≤ 6.1)+0.408 × age. According to Youden's criterion, the test is considered positive if pˆ > 9.63%, with being the estimated probability of AMH ≤ 1.1 ng/mL, with a sensitivity of 92% and a specificity of 78%. If the prevalence of having an AMH below 1.1 ng/mL is the one found in the sample used, 19%, the positive predictive value would be 50% (95% CI 36.6; 63.4) and the negative predictive value would be 97.6% (95% CI 85.9; 99.6). The latter result would indicate that this model is better for ruling out AMH levels (if, pˆ > 9.63%,97.6% have AMH greater than 1.1 ng/mL) than for confirming them.
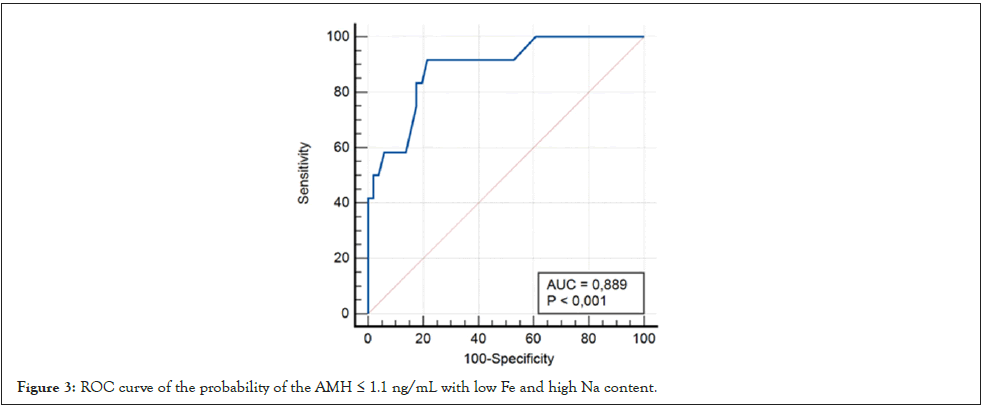
Therefore, low Fe or high Na levels had a significant relationship in the low AMH participants (Figure 3).
Figure 3: ROC curve of the probability of the AMH ≤ 1.1 ng/mL with low Fe and high Na content.
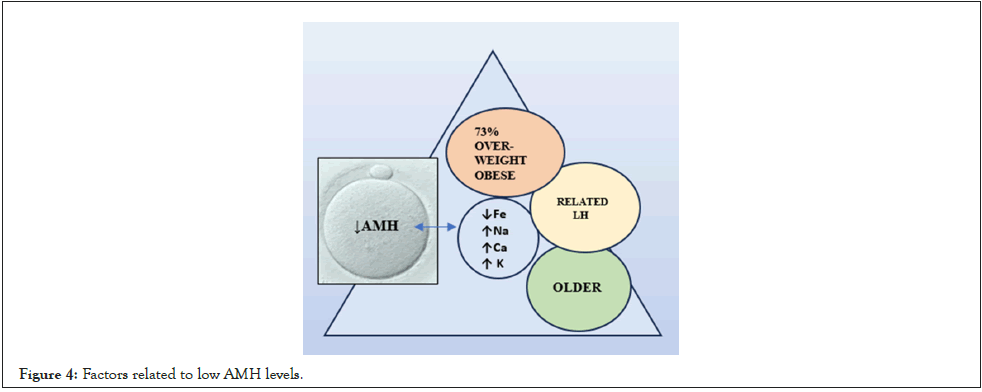
A relationship was established between low AMH and obesity, older age, relationship with LH, with an increase in Ca, Na, K and a decrease in Fe (Figure 4).
Figure 4: Factors related to low AMH levels.
Metals and FSH, LH, E2
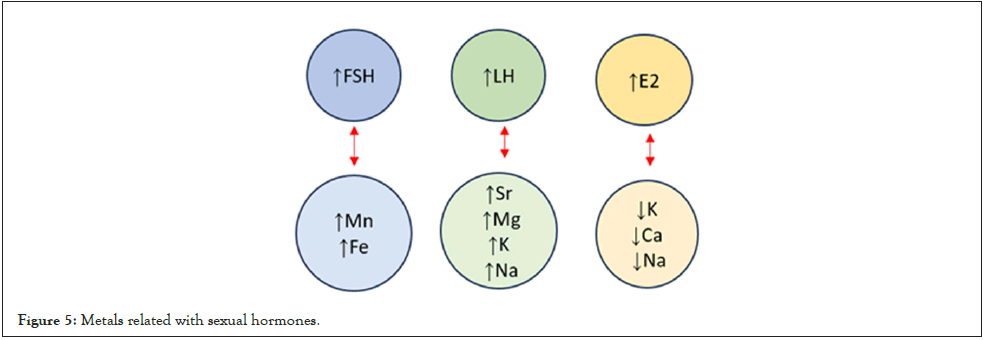
When FSH and metals were analyzed, a relationship was observed with Mn (rs=0.331, p=0.040) and this was close to significance with Fe (rs=0.229, p=0.081). There were only three women (4.8%) with values greater than 10 mUI/mL in whom values of Fe, Cu, Zn, and Al were detected, in addition to Ca, K, and Na.
In the case of LH and metals, there was a relationship with Sr (rs=0.303, p=0.020) and with Mg (rs=0.265, p=0.042) and this was close to significance with K (rs=0.228; p=0.083) and Na (rs =0.226, p=0.086).
Regarding E2, there was a relationship with K (rs=-0.395, p=0.003), Ca (rs=-0.305, p=0.023) and Na (rs=-0.323, p=0.016) (Figure 5).
Figure 5: Metals related with sexual hormones.
An inverse relationship was found between FSH levels and E2 (rs=-0.313, p=0.020) and a direct relationship between FSH and LH (rs=0.496, p<0.001). No significant relationship was observed with AMH and FSH (rs=0.123, p=0.352), nor between AMH and E2 (rs=-0.044, p=0.751), although there was a direct relationship between AMH and LH (rs=0.404, p=0.002).
Forty-nine women (78%) did not have PCO compared to fourteen (22%) who did. All participants with PCO had normal AMH levels (p<0.016), and the mean AMH levels were higher (3.6 ± 2.0, vs. 2.2 ± 1.5 p<0.007).
ART and AMH, FSH, LH, E2
The low AMH group had fewer oocytes (p<0.006), fewer mature oocytes (p<0.003), lower FR (p<0.003), lower cleavage rate (p<0.002), and lower Good Embryo Quality (GEQ) rate (p<0.003) than the normal AMH group.
Thirty-seven of the participants had a fresh embryo transfer (57.8%) and three women had a delayed transfer. Twenty-four of them (64.9%) had a single embryo transfer, and thirteen (35.1%) had a two-embryo transfer. Two participants had a second transfer with frozen embryos. No effect of AMH was found (p=0.685). Ten participants (27%) achieved implantation, and two of them were in the low AMH group. Ten of these embryos were successfully implanted, but two did not become pregnant (20%), both in the normal AMH group. There were eight evolutionary pregnancies. Two pregnancies were achieved in participants who had a second frozen embryo transfer, resulting in two newborns. The mean number of oocytes, mature oocytes, FR, CR, and GEQ were lower in the low AMH group (p<0.001).
FSH, LH, E2, and AMH were analysed and compared with FR, CR, GEQ, and Blastocyst Rate (BR) as continuous variables, and no relationship was noted between these variables.
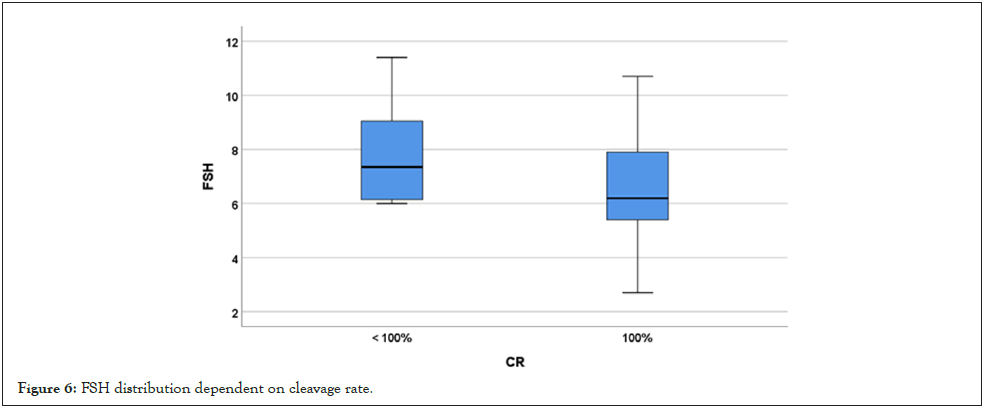
No association was seen with FR ≥ 75%, CR=100%, GEQ>66.6%, either with implantation or pregnancy. Thus, of the twenty women with FSH ≤ 5.9 mUI/mL, all of them (100%) had a 100% CR, compared to 78.4% (29 of 37) of those with FSH>5.9 mUI/mL (p=0.041) (Figure 6).
Figure 6: FSH distribution dependent on cleavage rate.
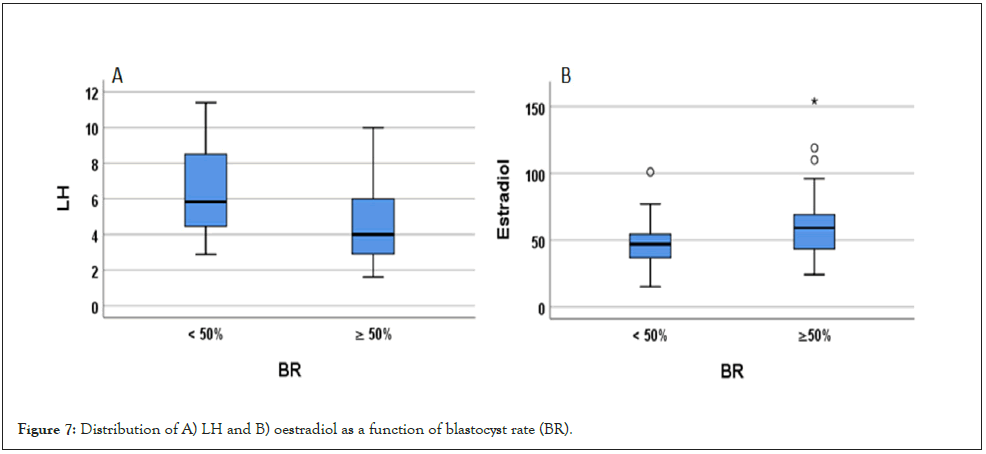
On the other hand, BR is significant for LH and close to significance for E2. The cut-off point according to Youden's criteria is LH ≤ 4.1 mUI/mL, with a sensitivity of 55.2% (CI95% 35.7; 73.6) and a specificity of 85.0% (CI95% 62.1;96.8). Among those with LH ≤ 4.1 mUI/mL, 84.2% had a BR ≥ 50%, whereas only 43.3% of those with LH>4.1 mUI/mL had a BR ≥ 50%, for the relationship between E2 and BR, the area under the ROC curve was 0.655 (p=0.058). The cut-off point according to Youden's criteria was E2>56 pg/mL, with a sensitivity of 53.6% (CI95% 33.9; 72.5) and a specificity of 84.2% (CI95% 60.4; 96.6). Of those with E2>56 pg/ mL, 83.3% had a BR ≥ 50%, whereas only 44.8% of those with E2 ≤ 56 pg/mL, had a BR ≥ 50% (p=0.014) (Figure 7).
Figure 7: Distribution of A) LH and B) oestradiol as a function of blastocyst rate (BR).
Discussion
As the FF content surrounds the developing oocyte, it is necessary to determine the elements present in it that may have some interaction. In the present study, the authors measured the metal content of FF to determine any influence on sexual hormones and consequently on ovarian reserve.
Metals and female sexual hormones
AMH: Chronic exposure to metals increases the incidence of low AMH levels by affecting ovarian function and impairing folliculogenesis [10]. Several authors reported that As, Mo, Sr, and V in FF were significantly increased in participants with low AMH, and may be a risk factor, while Cu and Mg levels were significantly decreased [28]. We found no differences in HM between patients with low or normal AMH levels. This may be due to the fact that there were only two participants with Pb. However, we also found no difference in Al, which was present in 70% of the participants, or Cu and Zn, which were present in almost 90% of the participants.
One of the main findings of this study is that Fe was significantly lower in the low AMH group, and the macroelements metal such as Ca, K and Na were significantly higher. Essential metals and macroelements play an important beneficial role in human health management, but, when their concentration exceeds regulatory limits, the resulting biotoxicity affects different organs and systems in biological structures [8]. This may be the reason why this study found a significant association between low Fe or high Na levels in participants with low AMH, suggesting a relevant influence of the macroelements Ca, K and Na and the essential metal Fe.
It was observed that Fe in FF was related to serum AMH levels, with significantly lower levels compared to infertile women from South China, there may be some difference in Fe exposure pathways. Hence, Fe may have a significant inverse effect on AMH, contrary to the results of the present study, probably due to some difference in Fe exposure pathways to these authors [17]. The relationship between Fe and AMH has been previously described in participants with transfusion-dependent ß-thalassemia [29].
Lower serum AMH was inversely related to serum ferritin levels, compared to similar healthy women, and the authors concluded that ovarian function may be impaired by chronic Fe overload [29].
In the present study, there were no women with such ovarian Fe overload, however, a low level was also considered to be associated with low serum AMH levels, implying that there are optimal levels of Fe above and below which ovarian function may be impaired. This means that Fe behaves as a ¨U shape¨ association between Fe concentration in FF and AMH levels as previously described [19]. Tian, et al. reported that the medium or the highest tertiles of Fe had significantly lower AMH, showing that higher concentrations enhance ovarian response, as we have found in our study [19]. This means that it is possible to supplement the diet with these elements Fe, to provide a more adequate environment for the growing follicle.
As is well known, AMH levels decrease with age, and low AMH levels are associated with a reduction in AF [30-32]. Thus, in the present study, the participants in the low group are significantly older, and there is a clear progressive decrease in AMH levels with increasing age. This is the result of a progressive depletion of the primordial follicular pool due to apoptosis [32].
Sometimes there is an increase in the permeability of the cations Ca+2, K+1 and Na+1 to the FF in participants with low AMH, as we observed in the present study. Furthermore, the movement of ions is involved in maintaining cellular stability under changing internal and environmental conditions and is an important part of the cell death process [32]. Therefore, could the increase in these metals be a consequence of AF apoptosis?
Na, K and Ca play important roles in physiological functions and in this sense are important elements that we found to be significantly associated with poor responders [19]. K is elevated in the FF of patients with DOR, providing the first evidence for the association between K and poor response, as we also found [19]. The excess of K accumulated in cells leads to changes in membrane potential, including Na accumulation, and contributes to the decline in fertility, as we found in our study group.
Ca is the most abundant inorganic element in the human body and one of its multiple functions is to act as a cofactor for numerous enzymes, as well as also to act in intracellular functions as a messenger in cascade signal reactions [33]. Apoptosis is a natural mechanism of programmed cell death, and Danese, et al. found that Ca2+ signalling plays a central role in triggering apoptosis in many cell types [34]. Given that low AMH levels reflect a decrease in the number of AF, the increase in Ca could be related to the destruction or apoptosis of AF. A higher concentration of intracellular Ca2+ has been observed in senescent cells [35].
Changes in ion levels, particularly K+, play a key role in the progression of apoptosis; K+ being the major intracellular ion is important for transition processes during the development of cell proliferative responses as well as the induction of programmed cell death via apoptosis. However, these authors found a decreased K level, unlike this study [34]. Apoptosis is accompanied by a change in the content of K+ and Na+ in cells and a decrease in the K/Na ratio [32].
Several studies have suggested that metals in the FF may play an important role in the disruption of hormone synthesis, so, the analysis of the association between metals and female hormones found increasing Cd levels and AMH [1,10]. However, in the present study, the participants did not have the same profile, due to the non-environmental or occupational exposure, therefore, we were not able to demonstrate these results.
E2: More than 70% of the studies in which E2 was analysed, had at least one metal with an inverse association [18]. With metal exposure, E2 synthesis and the levels of enzymes involved in ovarian steroidogenesis decrease, which could be inhibited by affecting the methylation levels of the Steroidogenic Factor-1 (SF-1) promoter region [28,36]. However, these studies do not analyse the macroelement metals and the hormones included in the present study.
Since low responders are associated with lower levels of E2, higher levels of K, Ca and Na associated with significant values were observed, as occurs with low AMH patients. Gerald, et al. demonstrated a potential relationship between exposure to endocrine-disrupting Cd and Pb and serum estrogen levels in US women, in addition other authors, obtained a negative association between Pb and E2, suggesting that exposure to metals may disrupt hormonal homeostasis in adults, particularly in older women [37,38]. In the present study, only two patients had Pb in the FF, and we did not obtain Cd as they had no occupational or environmental exposure to these metals, so we could not confirm this finding.
FSH/LH: We found an association between higher FSH with Fe and Mg, and higher LH with Sr, Mg, Na and K. It is known that there are three typical patterns of metal distribution between the BFB: Enriching, limiting and balancing, and Fe is one of the metals that is distributed in a limiting pattern from the blood to the FF. This selective permeability can be modulated by hormones [21].
Maintaining an optimal levels of Fe in the ovarian environment, promotes the normal ovarian response, oocyte maturation and ovarian developmental competence. Although other authors have not found significant correlations between LH, FSH and any metal [39-41].
The relationship we found between FSH and LH with these metals suggests that they may have an influence on ovarian ageing, in agreement with other authors who have found that changes in the hormone accelerate ovarian ageing [20].
BMI influence on AMH: Increased BMI was associated with lower AMH levels [30,42,43]. A trend towards significance was discovered in the present work in the low AMH group in relation to being overweight/obese. This study shows that in infertile participants, AMH is inversely correlated with BMI, especially in participants younger than 35 years of age, and with normal ovarian reserve. BMI seems to influence the decrease in serum AMH levels in women with increasing body size [43]. In this context, leptin has been shown to be directly related to fat density. In healthy individuals, leptin has been shown to influence LH and E2 levels, suggesting that it may have a distinct regulatory effect on hormones involved in reproductive processes [44].
The present study found that 73% of the participants in the low AMH group were overweight or obese, a finding that was close to significance when compared with the BMI in the normal AMH group. However, the fact that this finding was not significant may be due to the fact that BMI is usually set below 30 kg/m2 in the author´s reproduction unit. AMH may be a better biomarker for predicting DOR in IVF cycles than AFC or FSH [45]. In this study, no significant relationship was determined between AMH and FSH. AMH is a direct indicator of ovarian reserve and FSH is not as reliable.
Smoking influence
Several authors have shown that AMH is lower in smokers, and is related to the effect of environmental factors, endocrine disruptors, and metals on ovarian reserve, with an inverse relationship between exposure to air pollution and the serum levels of AMH [4,46]. The author did not find an association with smoking due to the low incidence of female smokers in the study. The same is true for the other hormones that were studied.
Markers of ovarian reserve
When analysing the relationship between the serum levels of the hormonal markers studied, AMH had a much higher predictability of ovarian reserve compared to LH, whereas other authors reported that AMH and serum LH, FSH, and E2 showed a positive correlation [47,48]. The lack of correlation between AMH, and FSH and E2 may explain why they are not good markers of ovarian reserve, as FSH and E2 can be influenced by factors outside the ovary, such as obesity or others. Wang et al. showed that metals were related with E2 and with FSH [20]. Similarly, the present study identified a relationship between this hormone and Mn and Fe, but the above authors reported a significant inverse association of serum E2 and Hg, and Pb, and a significative association with higher FSH levels and higher Pb levels, which was not the case in the present study, which could be related to the lack of environmental exposure. The association between metals and FSH and E2 suggested that may influence ovarian ageing [20].
Metals, female sexual hormones and ART
Metal exposure has been associated with reduced fecundity and fertility in couples using ART and may alter the homeostasis of hormones that control sexual maturation by binding to critical hormones and receptors [49]. Thus, the number of oocytes retrieved in ART, was positively correlated with AMH [50]. Other studies detected that AMH levels in infertile women decrease with age and that the number of oocytes retrieved in infertile women is positively correlated with AMH [51]. Several authors reported that environmental metals can cause a delay in the CR of the zygote into a blastocyst, associated with implantation failure or recurrent miscarriage [49].
It has been shown that ionic channels for Ca2+, K+ and Cl- are in the granulosa cells. The K+ channels in these cells appear to play a role in controlling the oocyte membrane potential [52]. As there are fewer antral follicles present in the ovary, less K is taken up by the ionic channels of the cumulus cells and higher levels of free K were detected in the analysed FF. In the same way, Na+ is the extracellular counterpart of K+, and both contributes to regulate the amount of water in extracellular space by homeostatic osmotic processes. In the group studied here, levels of K>287.91 mg/kg were associated with a low ovarian response.
Intracellular Ca2+ plays an important role in the regulation of oocyte processes in mammalian oocytes. Since cAMP maintains meiotic arrest, a transient release of endogenous Ca2+ can signal the resumption of meiosis [53]. Ca2+ is involved in the oocyte membrane potential of the gap junctions between the oocyte and the cumulus cells. Also, Ca is also involved in steroidogenesis and is required for FSH-induced cAMP and LH-induced progesterone production. However, Ca concentration does not reflect the status of the oocyte within the FF or its potential for fertilization. In fact, the results here showed a higher concentration of Ca in the FF of poor responders, but no differences were established in the FR, CR or GEQ rates.
Although the implantation rate does not differ between the two groups, low and normal AMH, the fact that more oocytes and more embryos are obtained means more ET, and this leads to a higher cumulative PR.
As expected, all the participants with PCO in the present study had AMH levels above 1.1 ng/ml [30,42]. This is consistent with the results obtained in these participants, with a higher number of oocytes, mature oocytes, FR, CR and GEQ.
The results here show that BR is significant for LH and almost significant for E2. Based on the studies of Chen et al. the LH Receptor (LH-R) mRNA transcription increased with the stage of embryo development, which would explain the relationship between LH and BR. On the other hand, Chang et al. demonstrated the expression of oestrogen receptor a (ERa) and oestrogen receptor b (ERb) in preimplantation stage embryos in vitro [54-55].
The present study, as well as studies by other authors, indicate the importance of avoiding heavy metal elements and the convenience of a correct supplementation of essential metals as well as an adequate diet to lead to an improved ovarian function.
Conclusion
This is the first study on the influence of metals on female sex hormones carried out in women in the Canary Islands. The study confirms the importance of metals regarding hormone levels.
A significant direct relationship was established between Mn in FF and FSH levels. A relationship close to statistical significance was observed between Fe levels and FSH. These findings suggest potential interactions between specific metals and FSH production or regulation.
Sr and Mg concentrations in FF were detected to be significantly related to LH levels. Relationships close to statistical significance were found between LH and K and Na levels. These findings suggest that certain metals may influence LH production or function in the female reproductive system.
An inverse relationship was observed between K, Ca, and Na concentrations in FF and E2 levels.
Lower Fe levels in FF presented significantly low AMH (≤ 1.1 ng/ mL), compared with normal AMH. Significantly higher levels of Ca and Na were obtained in the FF of women with low AMH. A non-significant trend towards higher K levels was identified in low AMH group. Low AMH was associated with older age and a trend toward higher BMI in the study participants.
In summary, the authors identified several parameters, weight, age, AMH levels, that can be associated with DOR, and this is the first study in which essential metal levels were included among these factors that can be related directly to low AMH.
The analysis of metals in FF opens a new line of research in the study of female sexual hormones. Future studies are needed to further investigate the possible association between metal exposure and female sex hormones and ovarian reserve. Understanding metal effects on hormones and ovarian reserve could have significant implications for reproductive health and ART. More research is required to investigate the association between metal exposure and female reproductive function in greater depth.
Limitations
When analysing the results obtained here, it is important to highlight the limitations of a relatively small sample size of 63 participants such as that of this study. The small sample size undoubtedly affects the representativeness of the results and the ability to generalize the findings to a larger population. The power analysis study carried out setting an accuracy of one standard deviation shows 81.1%, which could be somewhat lower if the accuracy were increased, for example, to half a standard deviation.
These results should be interpreted with caution. Future studies should be conducted with a larger sample size, which would provide more robust results and thus a better interpretation of the results. Despite the inherent limitations of the sample size, the results obtained provide a solid basis for future research in this area. It would also be possible to explore the relevant aspects in greater depth, which could contribute to a more complete understanding of the issue.
The present study was conducted in a small geographical area with participants of similar ethnicity. Therefore, the results of the study cannot be extrapolated to other ethnic groups or women living in different regions.
Conflict of Interest
The authors declare that they have no competing interests.
Ethical Standards
The authors declare that all procedures contributing to this work complied with the ethical standards of the relevant national and institutional committees on human experimentation and with the Helsinki Declaration of 1975, as revised in 2008. The present study protocol was reviewed and approved by the Institutional Review Board of the Hospital Universitario de Canarias (Reg. No. CHUC 2018 53), and all eligible participants signed an informed consent form prior to enrollment.
Author’s Contribution
Rubí Rodríguez-Díaz: Conceptualization, Methodology, Investigation, Original draft preparation, Visualization. Raquel Blanes-Zamora: Data curation, original draft preparation, Investigation. BS, Jorge Gómez-Rodríguez, Soraya Paz-Montelongo, Jonay González-Pérez, Carmen Rubio, Ángel J. Gutiérrez, Samuel Alejando Vega: Visualization, Investigation, Arturo Hardisson: Supervision, Validation, Resources. Enrique González-Dávila: Software, Validation, Methodology. All authors: Writing- Reviewing and Editing.
Data Availability
The data supporting the results of this study are available from the corresponding author on reasonable request.
References
- Kim K, Pollack A, Nobles C, Sjaarda L, Zolton J, Radoc J, et al. Associations between blood cadmium and endocrine features related to PCOS-phenotypes in healthy women of reproductive age: A prospective cohort study. Environ Health. 2021;20(1):64.
[Crossref] [Google Scholar] [PubMed]
- Du Y, Guo N, Wang Y, Teng X, Hua X, Deng T, et al. Follicular fluid concentrations of phthalate metabolites are associated with altered intrafollicular reproductive hormones in women undergoing in vitro fertilization. Fertil Steril 2019;111(5): 953-961.
[Crossref] [Google Scholar] [PubMed]
- Liu D, Shi Q, Liu C, Sun Q, Zeng X. Effects of endocrine-disrupting heavy metals on human. Health Toxics 2019;11(4):322.
[Crossref] [Google Scholar] [PubMed]
- Abareshi F, Sharifi Z, Hekmatshoar R, Fallahi M, Najafib M, Asour A, et al. Association of exposure to air pollution and green space with ovarian reserve hormones levels. Environ Res. 2020;184:109342.
[Crossref] [Google Scholar] [PubMed]
- Yan Y, Guo F, Liu K, Ding R, Wang Y. The effect of endocrine-disrupting chemicals on placental development. Front Endocrinol. 2023;14:105984.
[Crossref] [Google Scholar] [PubMed]
- Guo Q, Zhan Y, Li Y, Hong N, Guan Y, Zhang Z, et al. Investigating toxicity of urban road deposited sediments using Chinese hamster ovary cells and chlorella pyrenoidosa. Chemosphere. 2020;245:125634.
[Crossref] [Google Scholar] [PubMed]
- Singh R, Gautam N, Mishra A, Gupta R. Heavy metals and living systems: An overview. Indian J Pharmacol. 2011;43(3):246.
[Crossref] [Google Scholar] [PubMed]
- Briffa J, Sinagra E, Blundell R. Heavy metal pollution in the environment and their toxicological effects on humans. Heliyon. 2020;6(9):e04691.
[Crossref] [Google Scholar] [PubMed]
- ScutaraÈ?u EC, TrincÄ? LC. Heavy metals in foods and beverages: Global situation, health risks and reduction methods. Foods. 2023;12(18):3340.
[Crossref] [Google Scholar] (All versions) [PubMed]
- Wafa Y, Taha E, Nar A, Ateia A. Correlation between plasma lead and serum anti mullerian hormone levels in women with premature ovarian failure. Al-Azhar. Med J. 2020;49(2):469-480.
- Chen J, Ahn KC, Gee NA, Gee SJ, Hammock BD, Lasley BL. Antiandrogenic properties of parabens and other phenolic containing small molecules in personal care products. Toxicol Appl Pharmacol. 2007;221(3):278-84.
[Crossref] [Google Scholar] [PubMed]
- Tian T, Hao Y, Wang Y, Xu X, Long X, Yan L, et al. Mixed and single effects of endocrine disrupting chemicals in follicular fluid on likelihood of diminished ovarian reserve: A case-control study. Chemosphere. 2023;330:138727.
[Crossref] [Google Scholar] [PubMed]
- Devine K, Mumford SL, Wu M, De Cherney AH, Hill MJ, Propst A. Diminished ovarian reserve in the United States assisted reproductive technology population: Diagnostic trends among 181,536 cycles from the society for assisted reproductive technology clinic outcomes reporting system. Fertil Steril. 2015;104(3):612-19.e3.
[Crossref] [Google Scholar] [PubMed]
- Ferraretti A, Gianaroli L. The Bologna criteria for the definition of poor ovarian responders: Is there a need for revision? Hum Reprod. 2014;9(1):1842-1845.2022.
[Crossref] [Google Scholar] [PubMed]
- Granger E, Tal R. Anti-Müllerian hormone and its predictive utility in assisted reproductive technologies outcomes. Clin Obstet Gynecol. 2019;62(2):238-256.
[Crossref] [Google Scholar] [PubMed]
- Lee S, Min J, Min K. Female infertility associated with blood lead and cadmium levels. Int J Environ Res Public Health. 2020;17(5):1794.
[Crossref] [Google Scholar] [PubMed]
- Xu J, Ying Y, Chen J, Li D, Zhang D. Regional difference of metal level in follicular fluid and serum AMH level between infertile women form eastern China and southern China. Fertil Steril. 2019;112:3.
- Rami Y, Ebrahimpour K, Maghami M, Shoshtari-Yeganeh B, Kelishadi R. The association between heavy metals exposure and sex hormones: A systematic review on current evidence. Biol Trace Elem Res. 2022;200(8):3491-3510.
[Crossref] [Google Scholar] [PubMed]
- Tian T, Wang Z, Liu F, Fu Yu, Kong F, Wang Y, et al. Exposure to heavy metallic and trace essential elements and risk of diminished ovarian reserve in reproductive age women: A case-control study. J Hazard Mater. 2024;470:134206.
[Crossref] [Google Scholar] [PubMed]
- Wang X, Ding N, Harlow SD, Randolph JF Jr, Mukherjee B, Gold EB, et al. Exposure to heavy metals and hormone levels in midlife women: The Study of Women's Health Across the Nation (SWAN). Environ Pollut. 2023;317:120740.
[Crossref] [Google Scholar] [PubMed]
- Zhang G, Lin W, Gao N, Lan C, Ren M, Yan L, et al. Using machine learning to construct the blood–follicle distribution models of various trace elements and explore the transport-related pathways with multiomics data. Environ Sci Technol. 2024;58(18):7743-7757.
[Crossref] [Google Scholar] [PubMed]
- Rodríguez-Díaz R, Blanes-Zamora R, Paz-Montelongo S, Gómez-Rodríguez J, Rodríguez-Fiestas S, González-Weller D, et al. The influence of follicular fluid metals on assisted reproduction outcome. Biol Trace Elem Res. 2023;1-14.
[Crossref] [Google Scholar] [PubMed]
- Husten C. How should we define light or intermittent smoking? Does it matter? Nicotine Tob Res. 2009;11:111–121.
[Crossref] [Google Scholar] [PubMed]
- Associations between blood cadmium and endocrine features related to PCOS-phenotypes in healthy women of reproductive age: A prospective cohort study
- Crossref
- De Mendoza MH, Cuadros J, Arroyo G, Ten J, Pons M, Prados F, et al. Criterios ASEBIR de valoración morfológica de oocitos, embriones tempranos y blastocistos humanos. 3rd ed. Cuadernos de Embriología Clínica, Madrid: GÓBALO Gráfica. 2015.
- Google Scholar
- Chen Y, Sun Y, Zhao A, Cai X, Yu A, Xu Q, et al. Arsenic exposure diminishes ovarian follicular reserve and induces abnormal steroidogenesis by DNA methylation. Ecotoxicol Environ Saf. 2022;241:113816.
[Crossref] [Google Scholar] [PubMed]
- Chang H, Chen M, Lu M, Chern J, Lu C, Yang Y, et al. Iron overload is associated with low anti-müllerian hormone in women with transfusion-dependent β-thalassaemia. BJOG. 2011;118:825–831.
[Crossref] [Google Scholar] [PubMed]
- Bhattacharya K, Saha I, Sen D, Bose C, Chaudhuri G, Dutta S, et al. Role of anti-mullerian hormone in polycystic ovary syndrome. Middle East Fertil Soc J. 2022;27:32.
- Gunasheela D, Murali R, Appaneravanda LC, Gerstl B, Kumar A, Sengeetha N, et al. Age-specific distribution of serum anti-mullerian hormone and antral follicle count in Indian infertile women. J Hum Reprod Sci. 2021;14:372-9.
[Crossref] [Google Scholar] (All versions) [PubMed]
- Marakhova I, Yurinskaya V, Domnina AP. The role of intracellular potassium in cell quiescence, proliferation, and death. Int J Mol Sci. 2024;25(2):884.
[Crossref] [Google Scholar] [PubMed]
- Zoroddu MA, Aaseth J, Crisponi G, Medici S, Peana M, Nurchi VM. The essential metals for humans: A brief overview. J Inorg Biochem. 2019;195:120-129.
[Crossref] [Google Scholar] [PubMed]
- Danese A, Patergnani S, Bonora, Wieckowski MR, Previati M, Giorgi C, et al. Calcium regulates cell death in cancer: Roles of the mitochondria and mitochondria-associated membranes (MAMs). Biochim Biophys Acta Bioenergy. 2017;1858(8):615-627.
[Crossref] [Google Scholar] [PubMed]
- Borodkina AV, Shatrova AN, Deryabin PI, Griukova AA, Abushik PA, Antonov SM, et al. Calcium alterations signal either to senescence or to autophagy induction in stem cells upon oxidative stress. Aging (Albany NY). 2016;8(12):3400-3418.
[Crossref] [Google Scholar] [PubMed]
- Yu H, Kuang M, Wang Y, Rodeni S, Wei Q, Wang W, et al. Sodium arsenite injection induces ovarian oxidative stress and affects steroidogenesis in rats. Biol Trace Elem Res. 2019;189(1):186-193.
[Crossref] [Google Scholar] [PubMed]
- Gerald A, Ganapathy S, Zhu J, Wei Y. Exposure to endocrine-disrupting metals and serum estrogen levels among US women. Reprod Toxicol. 2023;118:108392.
[Crossref] [Google Scholar] [PubMed]
- Liu Q, Hu S, Fan F, Zheng Z, Zhou X, Zhang Y. Association of blood metals with serum sex hormones in adults: A cross-sectional study. Environ Sci Pollut Res Int. 2023;30(26):69628-69638.
[Crossref] [Google Scholar] [PubMed]
- Chen C, Wang N, Nie X, Han B, Li Q, Chen Y, et al. Blood cadmium level associates with lower testosterone and sex hormone-binding globulin in Chinese men: From SPECT-China study, 2014. Biol Trace Elem Res. 2016;171(1):71-78.
[Crossref] [Google Scholar] [PubMed]
- Pan W, Ye X, Zhu Z, Li C, Zhou J, Liu J. Urinary cadmium concentrations and risk of primary ovarian insufficiency in women: A case–control study. Environ Geochem Health. 2021;43(5):2025–2035.
[Crossref] [Google Scholar] [PubMed]
- Singh S, Kulshrestha R, Verma J, Nigam V, Kulshrestha M. Uncovering ovarian reserve: Insights into hormones, lipids, and essential metals. IJOGR. 2024;11(2):222-225.
- Butt M, Saleem J, Aiman S, Zakar R, Sadique I, Fischer F. Serum anti-Müllerian hormone as a predictor of polycystic ovarian syndrome among women of reproductive age. BMC Women’s Health. 2022;28 (22):199.
[Crossref] [Google Scholar] [PubMed]
- Albu D, Albu A. The relationship between anti-Müllerian hormone serum level and body mass index in a large cohort of infertile patients. Endocrine. 2019;63(1):157-163.
[Crossref] [Google Scholar] [PubMed]
- Imterat M, Agarwal A, Esteves S, Meyer J, Harlev A. Impact of body mass index on female fertility and ART outcomes. Panminerva Medica. 2019;61(1): 58-67.
[Crossref] [Google Scholar] [PubMed]
- Miyagi M, Mekaru K, Nakamura R, Oishi S, Akamine K, Heshiki C, et al. Live birth outcomes from IVF treatments in younger patients with low AMH. JBRA Assist Reprod. 2021;21(25):417-421.
[Crossref] [Google Scholar] (All versions) [PubMed]
- White AJ, Sandler DP, D’Aloisio AA, Stanczyk F, Whitworth K, Baird D, et al. Antimüllerian hormone in relation to tobacco and marijuana use and sources of indoor heating/cooking. Fertil Steril. 2016;106(3):723-730.
[Crossref] [Google Scholar] [PubMed]
- Wani S, Ajaz S, Rashid L, Ahmed J, Khurshid R, Ahmed A. Determination of ovarian reserve in different age groups of infertile women. J Sci Soc. 2020;47(3):164-167.
- Sinha S, Sharan A, Sinha S. Anti-mullerian hormone as a marker of ovarian reserve and function. Cureus. 2022;14 (9):e29214.
[Crossref] [Google Scholar] [PubMed]
- Obasi CN, Frazzoli C, Orisakwe OE (2022) Heavy metals and metalloids exposure and in vitro fertilization: Critical concerns in human reproductive medicine. Front Reprod Health. 2022;21(4)1037379.
[Crossref] [Google Scholar] [PubMed]
- Baker VL, Gracia C, Glassner MJ, Schnell VL, Doody K, Coddington CC, et al. Multicentre evaluation of the Access AMH antimüllerian hormone assay for the prediction of antral follicle count and poor ovarian response to controlled ovarian stimulation. Fertil Steril. 2018;110 (3):506-513.e3.
[Crossref] [Google Scholar] [PubMed]
- Sun XY, Lan YZ, Liu S, Long XP, Mao XG, Liu L. Relationship between anti-müllerian hormone and in vitro fertilization-embryo transfer in clinical pregnancy. Front Endocrinol (Lausanne). 2020;4(11):595448.
[Crossref] [Google Scholar] [PubMed]
- Arellano R, Martínez-Torres A, Garay E. Ionic currents activated via purinergic receptors in the cumulus cell-enclosed mouse oocyte. Biol Reprod. 2002;67(3):837–846.
[Crossref] [Google Scholar] [PubMed]
- Homa ST. Calcium and meiotic maturation of the mammalian oocyte. Mol Reprod Dev. 1995;40(1):122-134.
[Crossref] [Google Scholar] [PubMed]
- Chen X, Li J, Lu B, Qi Q, Zhang S, Zhuang G. LH-receptor messenger-RNA expression and LH secretion of the pre-implantation embryos correlated with human embryogenesis in vitro fertilization. Fertil Steril. 2013;100(3):S293-S294.
- Chang KT, Su YT, Tsai YR, Lan KC, Hsuuw YD, Kang HY, et al. High levels estradiol affect blastocyst implantation and post-implantation development directly in mice. Biomed. 2022;45(1):179-189.
[Crossref] [Google Scholar] [PubMed]
Citation: Gómez-Rodríguez J, Rodríguez-Díaz R, Blanes-Zamora R, Paz-Montelongo S, Vega SA, González-Pérez J, et al. (2025) Influence of Follicular Fluid Metals on Female Sexual Hormones. J Clin Med Sci. 9:304.
Copyright: © 2025 Gómez-Rodríguez J, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.