Indexed In
- Academic Journals Database
- Open J Gate
- Genamics JournalSeek
- JournalTOCs
- ResearchBible
- Ulrich's Periodicals Directory
- Electronic Journals Library
- RefSeek
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- Scholarsteer
- SWB online catalog
- Virtual Library of Biology (vifabio)
- Publons
- MIAR
- Geneva Foundation for Medical Education and Research
- Euro Pub
- Google Scholar
Useful Links
Share This Page
Journal Flyer

Open Access Journals
- Agri and Aquaculture
- Biochemistry
- Bioinformatics & Systems Biology
- Business & Management
- Chemistry
- Clinical Sciences
- Engineering
- Food & Nutrition
- General Science
- Genetics & Molecular Biology
- Immunology & Microbiology
- Medical Sciences
- Neuroscience & Psychology
- Nursing & Health Care
- Pharmaceutical Sciences
Mini Review - (2021) Volume 12, Issue 3
Expression of NUP62 in the Development of Ovarian Cancer
Chen Yong1, Chen Hongyu2, Huang Shirong3 and Fan Xiangqun4*2Department of Laboratory Medicine, Zhongwei Tech Innovation Medical Research Institute Co., Ltd, Beijing, China
3Department of Laboratory Medicine, Fujian Medical University, Fuzhou, China
4Department of Pathology, Fujian Medical University, Fuzhou, China
Received: 19-Mar-2021 Published: 09-Apr-2021, DOI: 10.35248/2153-0602.21.12.240
Abstract
Objective: This study aims at exploring the relationship between the universal differentially expressed genes and the development of ovarian cancer, and then we established a biological molecular model for the further biological study.
Methods: We selected the universal different expression gene, which have not yet been studied and with more than 60% coverage based on the 69 pairs of matched ovarian cancer and normal expression profiles samples with the directed network, and then analyzing the multidimensional data of NUP62 and miRNA-495.
Results: We found that mir-495, which target NUP62, was lower in patients with ovarian cancer.
Conclusion: The universal different expression of NUP62 is relevant to proliferation and apoptosis in ovarian cancer.
Keywords
NUP62; miRNA; Ovarian cancer; Expression gene; Malignant tumors; Gene expression; Adenocarcinoma
Introduction
Ovarian cancer is one of the most common malignant tumors because its morbidity and mortality are the highest in the world [1-3]. Adenocarcinoma is the most common tissue type of ovarian cancer. It has a high degree of malignancy and rapid development. Most of them are in the late stage when they are diagnosed. The median survival time is about one year, and the 5-year survival rate is less than 16%. The prognosis is poor, and the incidence is still increasing year by year. At present, the diagnosis of ovarian cancer mainly depends on pathological and cytological examination, which requires biopsy to obtain tissues, and is not easy to obtain repeatedly. However, relying on symptoms, signs and imaging diagnosis has greater limitations. Therefore, the use of tumor markers to assist ovarian cancer screening and clinical diagnosis has been paid more and more attention and recognition.
In this study, we first screened the differentially expressed genes at the population level of patients with ovarian cancer, and then combined multiple gene expression profile data sets of ovarian cancer relative to the normal matched tissues beside the cancer to identify the genes with a lower confidence limit of differential coverage of more than 60%. The differentially expressed gene NUP62 was screened out and its related action network was found by directed regulatory network. For further verification, we detected the expression level of NUP62 and related miRNA in ovarian cancer samples.
Literature Review
We selected the universal different expression gene, which have not yet been studied and with more than 60% coverage based on the 69 pairs of matched ovarian cancer and normal expression profiles samples with the directed network, and then analyzing the multidimensional data of NUP62 and miRNA-495
Data and preprocessing
Gene Expression Omnibus was used in this study. The methylation profiles of the matched ovarian cancer and its adjacent normal tissue promoters are all standardized series matrix files downloaded directly from gene expression omnibus.
Recognition of differentially expressed genes with high coverage
Firstly, the difference of expression value of each gene in ovarian cancer and its matched normal tissues beside the cancer was calculated, and the difference of expression value of all genes in each sample was ranked from small to large, and the rank divided by the total number of genes was taken as the difference score of expression value in this pair of samples.
Histological examination
In order to verify the clinical practicability of the difference of bioinformatics screening, we take the patients' tissue samples for left immunohistochemical staining and serological test.
Screening differentially expressed genes with high coverage in ovarian cancer
We control (False Discovery Rate) FDR <0.01. In all ovarian cancer samples, the frequency of these differentially expressed genes ranges from 60.25% to 75.42%, among which 523 differentially up-regulated genes and 89 differentially down-regulated genes cover more than 60% (Table 1).
| Gene ID | Symbol | Coverage rate | Lower confidence limit | Upper confidence limit | Degree of deviation scoring | Degree of deviation of patch |
|---|---|---|---|---|---|---|
| 1508 | CTSB | 0.9512 | 0.9358 | 0.9702 | 0.8908 | 29 |
| 7076 | TIMP1 | 0.9501 | 0.931 | 0.9022 | 0.9152 | 9 |
| 871 | SERPINH1 | 0.9525 | 0.9125 | 0.91423 | 0.92858 | 6 |
| 1277 | COL1A1 | 0.947 | 0.9225 | 0.9715 | 0.8914 | 28 |
| 7070 | THY1 | 0.947 | 0.9225 | 0.9715 | 0.9012 | 20 |
| 633 | NUP62 | 0.9342 | 0.9979 | 0.9619 | 0.9105 | 16 |
| 55127 | HEATR1 | 0.9315 | 0.9074 | 0.9616 | 0.808 | 409 |
Table 1: High coverage differential gene.
The relationship between high coverage differentially expressed genes and directed regulatory networks in ovarian cancer
The database of human network signal regulation includes activation, inhibition and physical interaction. In order to further refine the study, we filtered out some high coverage differentially expressed genes that were widely studied in cancer, and got four high coverage differentially expressed genes that were less studied or not studied in ovarian cancer (Table 2).
| Gene ID | Symbol | Coverage rate | Lower confidence limit | Upper confidence limit | Degree of deviation Scoring | Degree of deviation patch |
|---|---|---|---|---|---|---|
| 1871 | E2F3 | 0.9283 | 0.9001 | 0.9566 | 0.8187 | 311 |
| 4061 | LY6E | 0.9283 | 0.9001 | 0.9566 | 0.8642 | 75 |
| 5315 | PKM | 0.9252 | 0.9 | 0.954 | 0.8184 | 313 |
| 6696 | SPP1 | 0.9252 | 0.9 | 0.954 | 0.9055 | 18 |
Table 2: Regulatory genes of high coverage differential genes.
Then, these four high coverage differentially expressed genes, together with the selected group differentially expressed genes, were mapped into a directed regulatory network to ensure that each node is related to ovarian cancer, and a one-step and two-step regulatory neighborhood sub network of high coverage differentially expressed genes is obtained. Among them, NUP62 can directly activate the regulated differential expression gene, which can be down regulated and significantly inhibit cell proliferation, cell cycle arrest in G1/S phase and induce cell apoptosis. In ovarian cancer, NUP62 has a positive correlation with invasion depth, lymph node metastasis, distant metastasis, tumor differentiation and TMN stage (Figure 1).
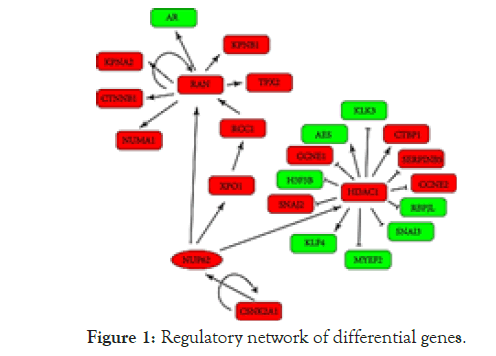
Figure 1: Regulatory network of differential genes
Multi-dimensional analysis of NUP62
In order to explore the reasons for the changes in the expression of NUP62, we calculated whether the miRNA of NUP62 regulated gene as the target gene changed (Table 3), and found that the hsa-mir-495 with NUP62 as the target gene had a copy number deletion. At the same time, the miRNAs targeting MAPK14 and CSNK2A1, the upstream regulatory genes of NUP62, also changed at the copy number level.
| GeneID | Symbol | Mir_name | Mir_ID | Copy number change | Ratio |
|---|---|---|---|---|---|
| 23636 | NUP62 | hsa-mir-101-2 | MI0000739 | Amplification Peak 16 | 14.85% |
| hsa-mir-495 | MI0003135 | Deletion Peak 35 | 31.21% | ||
| hsa-mir-888 | MI0005537 | Amplification Peak 36 | 19.22% | ||
| hsa-mir-924 | MI0005716 | Deletion Peak 42 | 31.04% | ||
| hsa-mir-509-3 | MI0005717 | Amplification Peak 36 | 28.82% |
Table 3: Changes of miRNA copy number related to differential expression of NUP62.
Discussion
The relatively normal differentially expressed genes in ovarian cancer indicated important information about the target of cancer occurrence, development and treatment [4]. Although the heterogeneity of ovarian cancer is very high, there will be a part of gene expression patterns in almost all ovarian cancer occurrence and development process, and this part of universally different expression genes may have potential significance for the occurrence, development and diagnosis and treatment of ovarian cancer. The relationship between NUP62 and ovarian cancer has not been studied. The high coverage genes we identified are still high coverage genes in different stages and subtypes. Based on the analysis of NUP62 gene with high coverage and differential expression, a biological molecular model was established, which provided a basis for further experimental verification. However, because the expression patterns of some genes are different in different stages or subtypes of ovarian cancer, the calculated coverage rate in all ovarian cancer samples does not represent the frequency of gene differential expression in a certain stage or subtype, so it is necessary to further classify ovarian cancer by stages and carry out corresponding analysis.
In view of the high heterogeneity of ovarian cancer patients and the complexity of cancer molecules, individualized diagnosis and treatment has also been promoted. In the process of occurrence and development of ovarian cancer, genes that are generally differentially expressed when compared with their own normal state suggest important information such as carcinogenesis mechanism and treatment target [5] which can be used as molecular markers. However, the coverage of differentially expressed genes in ovarian cancer is very small. In most studies, the single sample data set used for screening differentially expressed genes is small in sample size and poor in statistical efficiency due to the problems of laboratory funds, equipment and sample sources [6]. Moreover, due to the biological variation between individuals, the difference in the relative average reference value of gene expression does not mean the difference in the relative normal self [7].
Conclusion
Therefore, if we can integrate multiple data sets of matched samples of ovarian cancer relative to the adjacent cancer, and screen out the differential expression genes that do not depend on ovarian cancer subtypes and have high coverage, that is, the common molecular changes of ovarian cancer patients, it may provide new research ideas for exploring the mechanism of ovarian cancer occurrence and development, and have potential value for optimizing the diagnosis and treatment scheme of ovarian cancer.
Funding
This work was supported by the National Natural Science Foundation of Fujian Province (2017J01168). Fujian Provincial Key Laboratory sources played key supportive role for sample collection, molecular analysis of patient samples, bioinformatics analysis.
Conflict of Interests
The authors have no competing interests to declare.
Availability of Data and Materials
All data and materials generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Ethics Approval
The authors sincerely thank the participants for their help and willingness to take part in this study.
REFERENCES
- De Angelis R, Sant M, Coleman MP, Francisci S , Baili P, Pierannunzio D, et al. Cancer survival in Europe 1999-2007 by country and age: results of EUROCARE--5-a population-based study. Lancet Oncol. 2014;15(1): 23-34.
- Gansler T, Ganz PA, Grant M, Greene FL, Johnstone P, Mahoney M, et al. Sixty years of CA: a cancer journal for clinicians. CA Cancer J Clin. 2010;60(6): 345-50
- Siegel R, Ma J, Zou Z, Jemal A, Jemal A. CA: A cancer journal for clinicians 2014; CA Cancer J Clin 2014;64: 9-29.
- Vander Heiden MG, Cantley LC, Thompson CB. Understanding the Warburg effect: the metabolic requirements of cell proliferation. Science 2009; 324: 1029-1033.
- Wellen KE, Thompson CB. Cellular metabolic stress: considering how cells respond to nutrient excess. Mol Cell. 2010; 40(2): 323–332.
- Huang F, Ma B, Wang Y, Xiao R, Kong Y, Zhou X, et al. Targeting gene-virus-mediated manganese superoxide dismutase effectively suppresses tumor growth in hepatocellular carcinoma in vitro and in vivo. Cancer Biother Radiopharm. 2014;29(10): 403-11.
- Chatterjee A, Dasgupta S, Sidransky D. Mitochondrial subversion in cancer. Cancer Prev Res. 2011; 4: 638-654
Citation: Yong C, Hongyu C, Shirong H, Xiangqun F (2021) Expression of NUP62 in the Development of Ovarian Cancer. J Data Mining Genomics Proteomics.12:240
Copyright: © 2021 Yong C, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.