Indexed In
- Open J Gate
- Genamics JournalSeek
- JournalTOCs
- RefSeek
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- Publons
- Geneva Foundation for Medical Education and Research
- Google Scholar
Useful Links
Share This Page
Journal Flyer

Open Access Journals
- Agri and Aquaculture
- Biochemistry
- Bioinformatics & Systems Biology
- Business & Management
- Chemistry
- Clinical Sciences
- Engineering
- Food & Nutrition
- General Science
- Genetics & Molecular Biology
- Immunology & Microbiology
- Medical Sciences
- Neuroscience & Psychology
- Nursing & Health Care
- Pharmaceutical Sciences
Case Report - (2024) Volume 15, Issue 2
Convalescent Plasma for Critically-ill COVID-19 Patients
Adel A. Aljatham*, Waleed T. Hashim, Mohamad A. Almashiakhi, Abdulrahman M. Alharthy, Faisal A. Alaklobi, Mohammed Adam, Assem A. Elghazaly, Ahmed F. Mady, Daood S. Asad, Hala Aamr, Alaa H. Alali, Juhaina Abdlraheem, Muhned S. Alhumaid, Mohammed A. Aljatham and Khalid I. ZawbaeeReceived: 05-Feb-2024, Manuscript No. JCRB-24-24835; Editor assigned: 08-Feb-2024, Pre QC No. JCRB-24-24835 (PQ); Reviewed: 22-Feb-2024, QC No. JCRB-24-24835; Revised: 29-Feb-2024, Manuscript No. JCRB-24-24835 (R); Published: 08-Mar-2024, DOI: 10.35248/2155-9627.23.15.485
Abstract
Background and aim: Convalescent Plasma (CP) was early and successfully used in management of COVID-19 infection. This report aimed to document our experience with use of CP in two critically-ill COVID-19 patients.
Main findings: Patients presented with fever, dry cough and difficulty of breathing with reduced oxygen saturation, massive radiological lung involvement and deteriorated Sequential Organ Failure Assessment (SOFA) Score. Use of two sessions of CP resulted in marked improvement of radiological and/or pulmonary findings. Both patients died of septic shock or thromboembolic events.
Conclusion: Use of CP may be associated with radiological and clinical improvement in critically-ill patients. However, it’s not related to better survival if used in late stages of the disease.
Keywords
COVID-19; Passive immunization; Convalescent plasma
Introduction
The overwhelming challenge caused by COVID-19 pandemic triggered an unprecedented global effort to identify and develop effective medications and vaccines against the disease with different pathological targets, mechanisms, adverse effects and clinical outcomes [1]. Generally, these therapeutics include antiviral and immunomodulatory agents [2]. Among the immunomodulatory agents, passive antibody therapies including neutralizing monoclonal antibodies (mAbs), hyperimmunoglobulin and Convalescent Plasma (CP) have been early and successfully used [3]. In comparison with monocloncal antibodies, immune plasma from individuals immunized by both a natural SARS-CoV-2 infection and SARS-CoV-2 vaccination retained neutralizing activity against new variants [4].
Case Presentation
The first patient a 65-year-old male with history of hypertension. Patient presented to ER department in June, 2020 with 11-day history of fever, dry cough and difficulty of breathing. His peripheral oxygen saturation was 92.0% while receiving 15 L/min of oxygen via facemask. The patient was prompted to full laboratory assessment and radiological imaging. Laboratory findings are shown in Figure 1, Sequential Organ Failure Assessment (SOFA) Score was calculated to be 5 and chest radiograph revealed 70.0% of lung involvement with infiltrates. The patient was admitted to the ICU where he remained spontaneously breathing for 4 days. On deterioration of partial pressure of oxygen/fraction of inspired oxygen (P/F) ratio to 80, patient was switched to Mechanical Ventilation (MV) and maintained on full sedation and vasopressor support. During ICU stay, the patient received anti-viral medications, steroids, tocilizumab and multiple courses of antibiotics according to culture and sensitivity (ceftriaxone initially, followed by azithromycin, then linezolid and imipenem). In addition, patient received two sessions of CP (10-15 ml/kg infusion); the first at day 2 after initiation of MV and the second at day 4. CP administration resulted in marked improvement of chest radiographs to 50.0% and 40.0% of lung tissue involvement after the first and second sessions respectively. Also, P/F ratio improved to 500 at day 7 and tested negative for COVID-19 at day 13 of CP initiation. Unfortunately, however, the patient finally died after 45 days of ICU admission due to perforated acalculous cholecystitis, septic shock (main isolated organisms: Acinetobacter baumani and Staphylococcus capitis) and multi-organ failure.
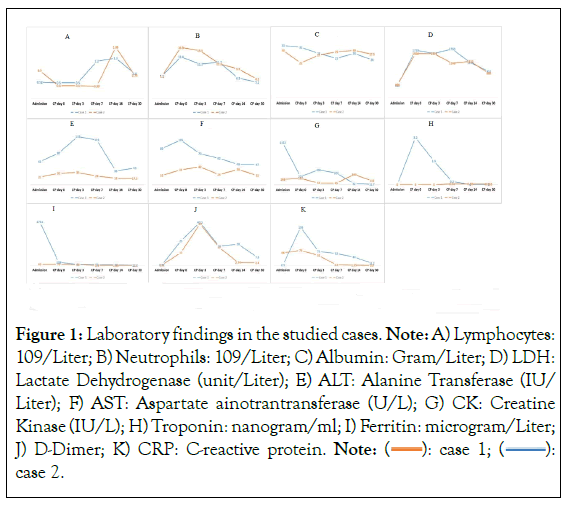
Figure 1: Laboratory findings in the studied cases. Note: A) Lymphocytes:
109/Liter; B) Neutrophils: 109/Liter; C) Albumin: Gram/Liter; D) LDH:
Lactate Dehydrogenase (unit/Liter); E) ALT: Alanine Transferase (IU/
Liter); F) AST: Aspartate ainotrantransferase (U/L); G) CK: Creatine
Kinase (IU/L); H) Troponin: nanogram/ml; I) Ferritin: microgram/Liter;
J) D-Dimer; K) CRP: C-reactive protein. Note:  case 2.
case 2.
The second patient is 68-year-old male with history of hypertension, diabetes, coronary heart disease, multiple disc herniations mainly L5-S1 and transient ischemic attacks. Upon presentation to ER in July, 2020, patient had complained of fever, dry cough, and difficulty of breathing for 3 days. Patient had a peripheral oxygen saturation of 91% while receiving 10 L/min of oxygen via facemask. Laboratory and radiological assessment identified a SOFA score of 4 and bilateral infiltration involving 40% of lungs fields. More laboratory details are illustrated in Figure 1.
The patient was admitted to ICU where he received anti-viral therapy, steroids, tocilizumab and empirical antibiotics (started with azithromycin and followed by complete courses of cefepime, vancomycin, and meropenim) due to lack positive cultures from any source. Three days after admission, radiological lung involvement extended to involve 80.0% of lung tissue. On the same day CP administration was initiated and the patient received two sessions. Radiological involvement was reduced to 50.0% and 40.0% of lung tissue at days 14 and 30 after CP use and COVID-19 test turned negative after 15 days of ICU admission. In spite of this improvement, the patient ultimately died after 93 days of ICU admission probably due to thromboembolic events.
Discussion
The present case report illustrates the clinical scenarios of two critically-ill COVID-19 patients who received CP during their ICU stay. While CP administration resulted in marked improvement of radiological findings and/or pulmonary functions, both patients finally died. The unfavorable outcome in our patients is probably attributed to the late use of CP. At this time, the patient usually has developed his own immune response which-if exaggerated- can result in cytokine storm and its consequences. Unfortunately, at the early months of the pandemic, no standardized protocols were available for CP administration.
This suggestion is supported by the conclusions of a recent meta- analysis of 58 randomized controlled trials. It was found that efficacy of CP is better when used in early disease stages [5].
In fact, the value of CP in management of COVID-19 patients is a controversial issue. One meta-analysis dedicated for studies involving outpatient COVID-19 patients revealed that patients who received CP, experienced lower rate of hospitalization with no benefit on survival or clinical outcome [6]. In contrast, another meta-analysis including the same subset of patients found that CP use was able to reduce all-cause mortality particularly when used within 5 days of symptom onset [7].
In hospitalized patients, use of CP resulted in reduction of mortality rate especially when preparations with high antibody levels were used or when CP was used early after hospitalization [8]. However, one case series acknowledged the efficacy and safety of CP even in late-state and severe cases of COVID-19 infection with significant improvement of clinical and laboratory findings and final discharge of 75.0% of patients [9].
In contrast, other studies concluded that CP use has no effect on mortality principally in patients with moderate-to-severe illness [10-12]. This controversy may be explained by the variable clinical characteristics of the studied patients and the variable preparations and protocols of CP administration in different centers.
This explanation is supported by the conclusions of Casadevall et al. who identified three conditions for successful use of CP in COVID-19 patients: 1) Use of specific antibody, 2) early use and 3) sufficient amount of antibodies [13].
Conclusion
Convalescent Plasma (CP) therapy has been utilized in the treatment of critically ill COVID-19 patients, resulting in marked improvement in radiological findings and/or pulmonary functions. However, both cases discussed ultimately ended in fatalities, possibly due to the late initiation of CP therapy. The timing of CP administration seems crucial, as indicated by studies showing better efficacy when used in early disease stages.
References
- Aboul-Fotouh S, Mahmoud AN, Elnahas EM, Habib MZ, Abdelraouf SM. What are the current anti-COVID-19 drugs? From traditional to smart molecular mechanisms. Virol J. 2023; 20(1):241.
[Crossref] [Google Scholar] [PubMed]
- Andrews HS, Herman JD, Gandhi RT. Treatments for COVID-19. Annu Rev Med. 2024; 75:145-157.
[Crossref] [Google Scholar] [PubMed]
- Yang X. Passive antibody therapy in emerging infectious diseases. Front Med. 2023;1-8.
[Crossref] [Google Scholar] [PubMed]
- Schrezenmeier H, Hoffmann S, Hofmann H, Appl T, Jahrsdorfer B, Seifried E, et al. Immune Plasma for the Treatment of COVID-19: Lessons Learned so far. Hamostaseologie. 2023; 43(1):67-74.
[Crossref] [Google Scholar] [PubMed]
- Stadler E, Chai KL, Schlub TE, Cromer D, Polizzotto MN, Kent SJ, et al. Determinants of passive antibody efficacy in SARS-CoV-2 infection: A systematic review and meta-analysis. Lancet Microbe. 2023; e883-e892.
[Crossref] [Google Scholar] [PubMed]
- Filippatos C, Ntanasis-Stathopoulos I, Sekeri K, Ntanasis-Stathopoulos A, Gavriatopoulou M, Psaltopoulou T, et al. Convalescent plasma therapy for COVID-19: A systematic review and meta-analysis on randomized controlled trials. Viruses. 2023;15(3):765.
[Crossref] [Google Scholar] [PubMed]
- Levine AC, Fukuta Y, Huaman MA, Ou J, Meisenberg BR, Patel B, et al. Coronavirus disease 2019 convalescent plasma outpatient therapy to prevent outpatient hospitalization: A meta-analysis of individual participant data from 5 randomized trials. Clin Infect Dis. 2023;76(12):2077-2086.
[Crossref] [Google Scholar] [PubMed]
- Senefeld JW, Gorman EK, Johnson PW, Moir ME, Klassen SA, Carter RE, et al. Rates among hospitalized patients with COVID-19 treated with convalescent plasma: A systematic review and meta-analysis. Mayo Clin Proc Innov Qual Outcomes. 2023; 7(5):499-513.
- Kumar NR, Karanam VC, Kumar S, Kumar SD. Convalescent plasma therapy in late-state, severe COVID-19 infection. South Med J. 2023;116(5):427.
[Crossref] [Google Scholar] [PubMed]
- Mihalek N, Radovanovic D, Barak O, Colovic P, Huber M, Erdoes G. Convalescent plasma and all-cause mortality of COVID-19 patients: Systematic review and meta-analysis. Sci Rep. 2023;13(1):12904.
[Crossref] [Google Scholar] [PubMed]
- Hakim SM, Chikhouni GM, Ammar MA, Amer AM. Effect of convalescent plasma transfusion on outcomes of coronavirus disease 2019: A meta-analysis with trial sequential analysis. J Anesth. 2023:1-4.
[Crossref] [Google Scholar] [PubMed]
- Iannizzi C, Chai KL, Piechotta V, Valk SJ, Kimber C, Monsef I, et al. Convalescent plasma for people with COVID‐19: A living systematic review. Cochrane Database Syst Rev. 2023(2).
[Crossref] [Google Scholar] [PubMed]
- Casadevall A, Joyner MJ, Pirofski LA, Senefeld JW, Shoham S, Sullivan D, et al. Convalescent plasma therapy in COVID-19: Unravelling the data using the principles of antibody therapy. Expert Rev Respir Med. 2023;17(5):381-395.
[Crossref] [Google Scholar] [PubMed]
Citation: Aljatham AA, Hashim WT, Almashiakhi MA, Alharthy AM, Alaklobi FA, Adam M, et al. (2024) Convalescent Plasma for Critically-ill COVID-19 Patients. J Clin Res Bioeth. 15:485.
Copyright: © 2024 Aljatham AA, et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

