Indexed In
- Open J Gate
- JournalTOCs
- The Global Impact Factor (GIF)
- RefSeek
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- Publons
- Euro Pub
- Google Scholar
Useful Links
Share This Page
Journal Flyer

Open Access Journals
- Agri and Aquaculture
- Biochemistry
- Bioinformatics & Systems Biology
- Business & Management
- Chemistry
- Clinical Sciences
- Engineering
- Food & Nutrition
- General Science
- Genetics & Molecular Biology
- Immunology & Microbiology
- Medical Sciences
- Neuroscience & Psychology
- Nursing & Health Care
- Pharmaceutical Sciences
Research - (2021) Volume 9, Issue 1
An Observational Study Report on Adverse Drug Reactions of A Combination of Drugs in A Tertiary Care Hospital, Kolkata, West Bengal
Sarbani Biswas1,2, Avijit Chatterjee1, Shyamshree S S Manna1 and Uddipan Kar3*2Jakir Hossain Institute of Pharmacy, Raghunathganj, West Bengal, India
3Massachusetts Institute of Technology, Cambridge, Massachusetts, USA
Received: 04-Jan-2021 Published: 25-Jan-2021, DOI: 10.35248/2329-6887.21.9.297
Abstract
Background: Drugs or proper medications can be a preventive measure against several temporary or chronic human diseases and physiological conditions. But sometimes, drugs can turn out to be a bane to human life rather than becoming a boon. Administration of certain drugs may lead to several undesirable detrimental effects to human health that may even be fatal if gone unnoticed. These reactions are more commonly referred to as the Adverse drug Reactionsâ™.
Aim: In the current study, our major aim is to probe the adverse drug reactions of two particular drugs Paracetamol and Cephalosporin, when administered in combination or separately in a tertiary care hospital, Kolkata, West Bengal.
Material and methods: Our study involves a hospital-based observational study where the adverse drug reactions reported by medical practitioners over a period of six months were assessed in patients administered with paracetamol and cephalosporin group of drugs in combination or separately.
Results: In this study, 100 patients were administered paracetamol and cephalosporin drug groups both separately or in combination. Out of them, 36 patients developed ADR like hepatotoxicity, hypotension, anemia, vomiting, skin rashes as well as Steven Johnson Syndrome. Adverse drug reaction is found to be more common in women than in men. Also, middle-aged adults (15-65 years) are more prone to adverse drug reactions on the administration of a combination of paracetamol and cephalosporin drugs. Further, when the patients were subjected to a dose of a combination of paracetamol and cephalosporin drug groups, they experienced mainly vomiting along with some minor cases of hypertension, hepatotoxicity, and skin rashes.
Conclusion: Our study clearly indicates that when a patient is co-administered a combination of both paracetamol and cephalosporin drug groups, the majority of the observed adverse drug reaction symptom is vomiting which is a signature adverse drug reaction symptom of cephalosporin drug group alone, indicating a clear predominance in symptoms of adverse drug reaction exhibited by cephalosporin drug group over paracetamol.
Keywords
Drugs; Adverse drug reactions; Paracetamol; Cephalosporins
Introduction
In present-day to day life, human beings are completely dependent on drugs and medicines for better overall health. Drugs can give relief to several temporary as well as chronic long-standing health issues in human life. But in reality, the same drugs which can save millions of lives can lead to severe side effects or adverse reactions in several patients which can sometimes even lead to death [1,2]. According to a recent report, adverse drug reactions turned out to be the 4th major cause of morbidity leading to deaths of about 2.5 lakhs of people every year [3]. One of the most wellknown cases of ‘Adverse drug reaction’ was the adverse effects of Thalidomide [4,5]. Thalidomide captured the drug market as an attractive sedative and anti-emetic drug used for the treatment of morning sickness experienced by pregnant ladies. But soon, the drug was found to be the causative agent of peripheral neuropathy in patients as well as certain birth defects in newborn babies like Phocomelia [6]. Another notable example of adverse drug reaction includes the withdrawal of a non-steroidal anti-inflammatory drug (NSAID) named Bromfenac due to reports of severe liver failure on its administration [7]. These incidents clearly throw light on the importance of the detection of adverse drug reactions among current circulating drugs. But still, not many past research studies are available on the topic.
A well-known highly consumable non-steroidal anti-inflammatory non-prescription drug in the present-day market is paracetamol [8,9], which is readily available to the general public. Although paracetamol is a potent antipyretic drug within the therapeutic dose, there were several reports linked to adverse effects of paracetamol in several patients [10,11]. The first reported case of adverse drug reaction of paracetamol dates back to 1966 when an overdose of the drug caused centrilobular hepatic necrosis that finally leads to renal necrosis [12,13]. There were also some reports, where an overdose of paracetamol lead to anaphylaxis reaction like hypotension [14,15] and hemodynamic/anemia [16].
Further, the number of research studies focused on the adverse drug reaction on antibiotics is very less. One of the most commonly used antibiotics is Cephalosporin [17-20]. Cephalosporin is known to show adverse drug reactions to certain patients [21]. According to recent reports, patients allergic to penicillin are more prone to adverse drug reaction to cephalosporin [22,23]. The most common adverse drug reactions to the cephalosporin group of drugs like Cefadroxil and Cefuroxime include urticaria, angioedema, anaphylaxis as well as bronchospasm [24]. In rare cases, Cefadroxil may lead to Steven Johnson Syndrome and exfoliative dermatitis [25].
In the current study, we have performed an observational-based study on the adverse drug reactions in patients administered with paracetamol and cephalosporin drugs separately as well as in combination. Adverse drug reactions are observed in 36 percent of the patients. Females are found to be more affected by adverse drug reactions as compared to males. Adult people (15-65) are found to be the high-risk group for adverse reactions to the combination of drugs. The major aim of the study was to identify the frequency and symptoms of adverse drug reactions related to common drugs like paracetamol and cephalosporin groups when prescribed alone or in combination. The effect of adverse drug reaction displayed by the cephalosporin drug group alone i.e. vomiting was found to be predominant over the symptoms of adverse drug reaction of paracetamol drug in majority of patients when both the drugs were administered simultaneously.
Materials and Methods
Study Design
The following study was an observational study conducted at R G Kar Medical College and Hospital, a tertiary care hospital in Kolkata, West Bengal, and India over a period of six months. A total of 100 patients of all age groups and genders were considered for the study. Consent letter was collected from all the patients. Data collection was done using a predesigned case record form which included patient characteristics such as age, gender, diagnosis, as well as prescription characteristic such as the name of the drug, strength and dosage form, number of units dispensed upon (based on their prescription or prescriber record). All biological tests, physical examinations, and other analytical test reports were recorded in detail. The patients were first explained all the adverse drug reactions and was advised to fill up the Central Drug Standard Control Organisation (CDSCO) adverse drug monitoring form as a record. The study was approved by the hospital’s Ethical Committee.
Drugs Administered
The patients were administered with paracetamol drug and drugs belonging to cephalosporin groups such as cefadroxil/cefuroxime were administered either separately or in combination.
Probability Algorithms
The probability of adverse drug reaction due to drug administration was evaluated by the Naranjo scale or Naranjo algorithm [26, 27] while assessment of the severity of reported adverse drug reaction was determined by the Hartwig severity assessment scale [28].
Definitions
Naranjo scale or Naranjo algorithm may be defined as a questionnaire that measures the probability of adverse drug reaction on the administration of drugs introduced first by Naranjo et al [26]. The algorithm was introduced to optimize casualty assessment for all adverse drug reactions Table 1. Based on the scores obtained from the assessment, the results are interpreted as doubtful adverse drug reaction (0), possible adverse drug reaction (1 – 4), probable adverse drug reaction (5 – 8), and definite adverse drug reaction (≥ 9) respectively.
Table 1A.| Question | Yes | No | Do Not know |
score |
|---|---|---|---|---|
| 1. Are there previous conclusive reports on this reaction? | +1 | 0 | 0 | |
| 2. Did the adverse event appear after the suspected drug was administered? |
+2 | -1 | 0 | |
| 3. Did the adverse reaction improve when the drug was dis-continued or a specific antagonist was administered? | +1 | 0 | 0 | |
| 4. Did the adverse reaction reappear when the drug was re administered? | +2 | -1 | 0 | |
| 5. Are there alternative causes (other than the drug) that could on their own have caused the reaction? |
-1 | +2 | 0 | |
| 6. Did the reaction reappear when a placebo was given? | -1 | +1 | 0 | |
| 7. Was the drug detected in the blood or other fluids in concentrations known to be toxic? | +1 | 0 | 0 | |
| 8. Was the reaction more severe when the dose was increased, or less severed when the dose was decreased |
+1 | 0 | 0 | |
| 9. Did the patient have a similar reaction to the same or similar drugs in any previous exposure? |
+1 | 0 | 0 | |
| 10. Was the adverse event confirmed by any objective evidence? | +1 | 0 | 0 | |
| Total score: | ||||
| Total Score | Probability of ADR |
|---|---|
| ≥ 9 | Definite |
| 5 to 8 | Probable |
| 1 to 4 | Possible |
| ≤ 0 | Doubtful |
Table 1: Table 1A shows the Naranjo assessment scale is a set of questions that determine the probability of adverse drug reactions in patients. Table 1B shows the probability of ADR based on scores from Table 1A.
Hartwig severity assessment scale is defined as an adverse drug reaction (ADR) severity assessment scale that categorizes ADR into three distinct classes: mild (1-2 points), moderate (3-4 points), and severe (5-7 points). The detailed description of the Hartwig scale is tabulated in Table 2.
| Severity | Descriptions |
|---|---|
| Mild | Self-limiting ADR reactions that can resolve over time without treatment and does not stay for long time |
| Moderate | ADR reactions that require therapeutic intervention and hospitalization prolong by 1 day but resolved in < 24 h or require a change in drug therapy or treatment procedure to check further worsening of the outcome. |
| Severe | ADR reactions that requires prolonged hospital stay and can pose serious threat to life sometimes may even be fatal. |
Table 2: Table depicts the Hartwig severity assessment scale.
Data Analysis
Several standard statistical procedures were used to analyse the data. The majority of the data are presented as a percentage and represented by corresponding bar diagrams.
Results
Demographic characteristics
The whole study was conducted with 100 patients. Out of 100 patients, 61patients were male and 39 patients were female (Figure 1). A total of 36 patients developed mild to severe several adverse drug reactions (ADR) out of 100. The patients, who developed adverse drug reactions were classified according to age groups such as pediatric (1-14 age group), adult (14-65 age group), and geriatric (65-95 age group) groups. Adults (83%) are found to be more prone to developing ADRs as compared to pediatric (7%) and geriatric (10%) age groups (Figure 2). The incidence of adverse drug reaction was higher in the case of females (34.42%) than males (38.46%) (Figure 3) The summary of the demographical characteristics of patients studied is tabulated in Table 3.
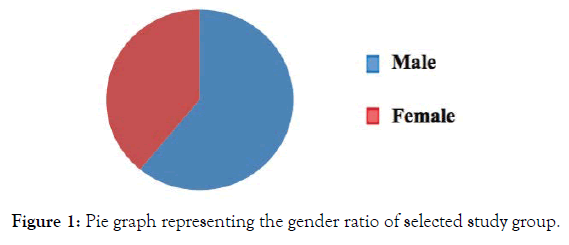
Figure 1. Pie graph representing the gender ratio of selected study group.
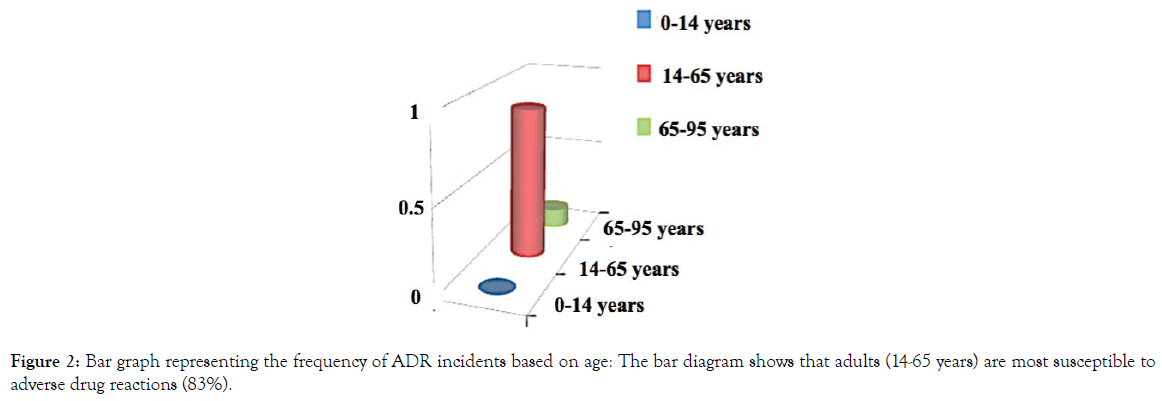
Figure 2. Bar graph representing the frequency of ADR incidents based on age: The bar diagram shows that adults (14-65 years) are most susceptible to adverse drug reactions (83%).
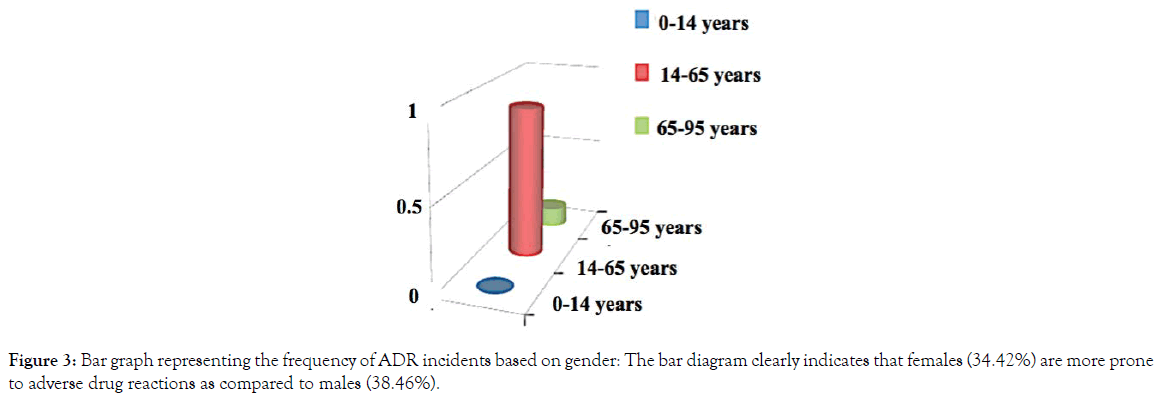
Figure 3. Bar graph representing the frequency of ADR incidents based on gender: The bar diagram clearly indicates that females (34.42%) are more prone to adverse drug reactions as compared to males (38.46%).
| Demography | Percentage of patients developed ADR (%) |
|---|---|
| Sex 1) Male 2) Female |
34.42 38.46 |
| Age 1) 0-14 years 2) 14-65 years 3) above 65 years |
7 83 10 |
Table 3: Table showing the characteristics of patients used in study (total patients studied (n)=100 patients).
Casualty and severity data
A detailed casualty assessment based on the Naranjo scale points out that almost 88.89% of adverse drug reactions (ADRs) were found to be possible while 8.33% ADRs were probable and the rest 2.78% ADRs were found definite (Figure 4). Hartwig’s severity assessment scale revealed that 86.11% of the reported ADRs were mild, 11.11% were moderate and 2.78% were severe (Figure 5).
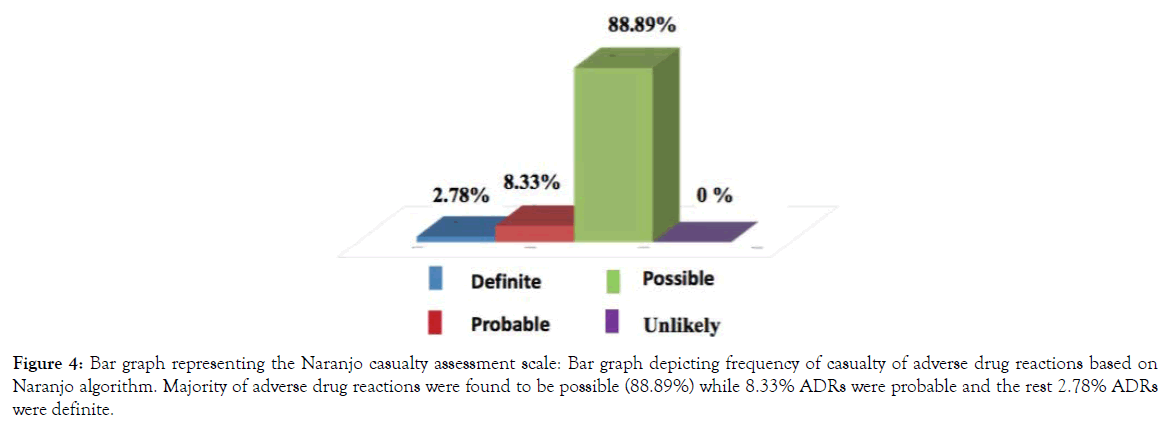
Figure 4. Bar graph representing the Naranjo casualty assessment scale: Bar graph depicting frequency of casualty of adverse drug reactions based on Naranjo algorithm. Majority of adverse drug reactions were found to be possible (88.89%) while 8.33% ADRs were probable and the rest 2.78% ADRs were definite.
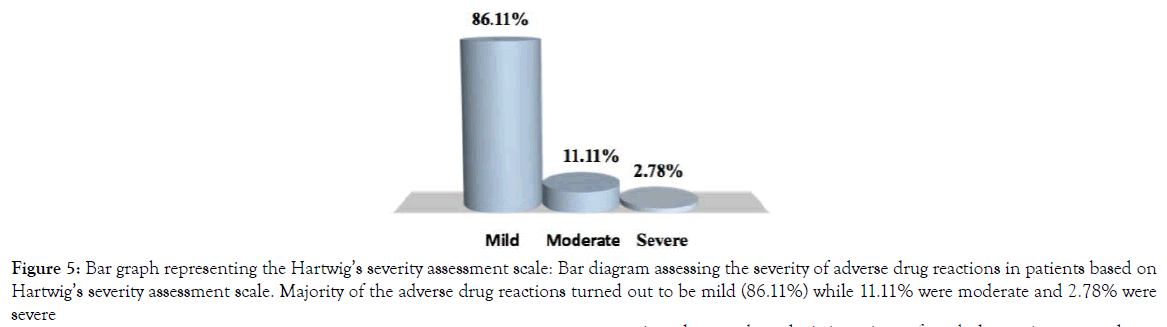
Figure 5. Bar graph representing the Hartwig’s severity assessment scale: Bar diagram assessing the severity of adverse drug reactions in patients based on Hartwig’s severity assessment scale. Majority of the adverse drug reactions turned out to be mild (86.11%) while 11.11% were moderate and 2.78% were severe
Drug related adverse drug reaction (ADR) frequency and symptoms
As previously mentioned, a total of 36 patients out of 100 developed adverse drug reactions (ADRs) The majority of reported adverse drug reactions were hepatotoxicity (36.11%) and hypotension (36.11%). Some of the other adverse drug reaction symptoms include vomiting (19.44%), anemia (2.78%), skin rash (2.78%) as well as Steven Johnson Syndrome (2.78%) (Figure 6) Administration of paracetamol alone leads to ADRs like hepatotoxicity, anemia, and hypotension. The major adverse drug reaction due to the administration of cephalosporin group drugs like cefuroxime and cefadroxil was vomiting.
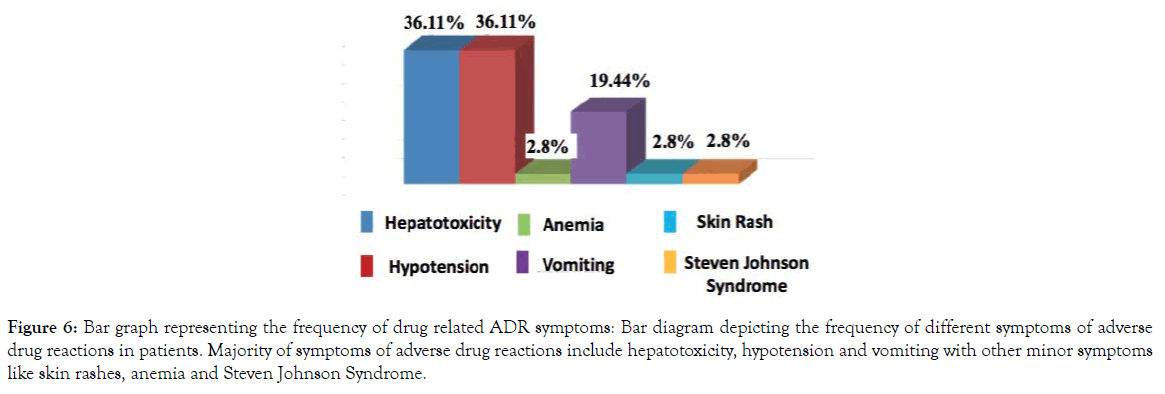
Figure 6. Bar graph representing the frequency of drug related ADR symptoms: Bar diagram depicting the frequency of different symptoms of adverse drug reactions in patients. Majority of symptoms of adverse drug reactions include hepatotoxicity, hypotension and vomiting with other minor symptoms like skin rashes, anemia and Steven Johnson Syndrome.
Administration of a combination of paracetamol and cephalosporin drug groups like cefuroxime and cefadroxil mainly leads to vomiting along with minor reports of hepatotoxicity, hypotension, anemia, skin rash, and Steven Johnson Syndrome. The detailed frequency of drug related ADRs are given in Table 4.
| Drugs Administered | ADR symptoms | Number of patients developing ADR based on symptoms |
|---|---|---|
| Paracetamol | 1. Hypotension 2. Hepatotoxicity 3. Anemia |
12 10 1 |
| Cephalosporin drug group | Vomiting | 2 |
| Paracetamol and cephalosporin both simultaneously | 1. Vomiting 2. Skin rashes 3. Hypotension 4. Hepatotoxicity 5. Steven Johnson’s syndrome |
6 1 1 2 1 |
Table 4: ADR symptoms and frequency when patients were administered with paracetamol and cephalosporin drug groups both separately and in combination.
Discussion
The whole study reveals in detail the frequency of casualty and severity of several adverse drug reactions (ADR) as well as their symptoms in hospitalized patients caused by commonly used lifesaving medications like paracetamol and cephalosporin drug groups when prescribed separately or in combination. Our study shows that 36 % of the patients developed adverse drug reactions. The relatively higher incidence of adverse drug reaction closely matches with the previous meta-analysis study by Lazarou et al, where they reported a 15.1 % ADR incidents among patients [29]. Similar results were shown by Miguel et al with ADR incidence reports of nearly 17% in hospitalized patients [30]. Although several other recent ADR studies revealed a lower incidence rate [31-33], the main reason behind that may be the spontaneous reporting systems used in those studies which lead to underreporting [34]. Our study further indicates the fact that the incidence of ADR is relatively higher in the case of females as compared to males which correlate well with previous reports [35]. Although the exact reason for this discrepancy remains unclear, this may be attributed to the basic physiological differences between males and females such as a difference in body mass index and fat composition as well as effects of hormones on the drug metabolism. Our study also shows that adults and older people were more susceptible to drug-related ADRs as compared to paediatric groups. One of the reasons behind that may be that aging leads to physiological changes that may alter drug pharmacokinetics (absorption, distribution, metabolism, and excretion) as well as pharmacodynamics (the study of effects of a drug on the body). Previous research also indicates a two-fold increase in ADR frequency in elderly people as compared to younger people [36-38]. Results from our study demonstrate that adverse drug reactions related to paracetamol include primarily hypertension and hepatotoxicity while the adverse drug reaction related to cephalosporin drug group leads to vomiting. In total, 23 patients experienced adverse drug reactions on paracetamol administration while only 2 patients experienced adverse drug reactions on cephalosporin drug group administration. Surprisingly, when the hospitalized patients were administered a combination of both paracetamol and cephalosporin drug groups, a majority of the patients (6 out of total 11 patients who developed adverse drug reactions) suffered from vomiting which was a signature symptom of cephalosporin drug administration alone while only a small group of people experienced other adverse drug reactions like hypertension, hepatotoxicity, vomiting, and skin rashes. Administration of combination of drug also leads to a rare adverse drug reaction like Steven Johnson Syndrome in a patient (Table 4). This clearly lays out the fact that there is a clear predominance in adverse drug reaction effects exhibited by the cephalosporin drug group over paracetamol when both the drugs are administered simultaneously. Further, the majority (88.89%) of the reported ADRs were turned out to be possible (as per Naranjo scale) while Hartwig severity assessment indicated most of the reported ADRs (almost 86.11%) were mild. One of the major limitations of the study was the use of the Naranjo scale or the Naranjo algorithm. Recent studies have raised questions on the use of the Naranjo algorithm which lead to the development of various new casualty assessment scales and tools like that of Liverpool ADR Casualty Assessment Tool (LCAT) [39]. In the future, we will aim to crosscheck the casualty data using the modified casualty assessment scales [40].
Conclusion
Our study gives result consistent with previous reports such as females are more prone to adverse drug reaction effects than male and established the fact that adults (between 14-65) are more susceptible to adverse drug reactions. Our study clearly provides detail symptoms of adverse drug reactions in patients administered with paracetamol and cephalosporin drug groups separately or in combination. Our study further confirms that when a patient is administered both paracetamol and cephalosporin drug groups simultaneously, majority of the observed adverse drug reaction case is vomiting which is a signature adverse drug reaction symptom of cephalosporin group alone.
REFERENCES
- Jones JK. Adverse drug reactions in the community health setting: approaches to recognizing, counseling, and reporting. Family & community health. 1982 Aug 1;5(2):58-67.
- Patidar D, Rajput MS, Nirmal NP, Savitri W. Implementation and evaluation of adverse drug reaction monitoring system in a tertiary care teaching hospital in Mumbai, India. Interdisciplinary toxicology. 2013 Mar 1;6(1):41-6.
- Hacker M. Adverse drug reactions. InPharmacology 2009 Jan 1 (pp. 327-352). Academic Press.
- Dimopoulos MA, Eleutherakis-Papaiakovou V. Adverse effects of thalidomide administration in patients with neoplastic diseases. The American journal of medicine. 2004 Oct 1;117(7):508-15.
- Ito T, Ando H, Handa H. Teratogenic effects of thalidomide: molecular mechanisms. Cellular and Molecular Life Sciences. 2011 May 1;68(9):1569-79.
- Vargesson N. The teratogenic effects of thalidomide on limbs. Journal of Hand Surgery (European Volume). 2019 Jan;44(1):88-95.
- Fontana RJ, McCashland TM, Benner KG, Appelman HD, Gunartanam NT, Wisecarver JL, et al, Acute Liver Failure Study Group. Acute liver failure associated with prolonged use of bromfenac leading to liver transplantation. Liver Transplantation and Surgery. 1999 Nov;5(6):480-4.
- Twycross R, Pace V, Mihalyo M, Wilcock A. Acetaminophen (paracetamol). Journal of pain and symptom management. 2013 Nov 1;46(5):747-55.
- Esh CJ, Mauger AR, Palfreeman RA, Al-Janubi H, Taylor L. Acetaminophen (paracetamol): use beyond pain management and dose variability. Frontiers in physiology. 2017 Dec 22;8:1092.
- Nagai J, Uesawa Y, Shimamura R, Kagaya H. Characterization of the adverse effects induced by acetaminophen and nonsteroidal anti-inflammatory drugs based on the analysis of the Japanese adverse drug event report database. The Clinical journal of pain. 2017 Aug;33(8):667.
- McCrae JC, Morrison EE, MacIntyre IM, Dear JW, Webb DJ. Long term adverse effects of paracetamol–a review. British journal of clinical pharmacology. 2018 Oct;84(10):2218-30.
- Davidson DG, Eastham W. Acute liver necrosis following overdose of paracetamol. British medical journal. 1966 Aug 27;2(5512):497.
- Boyd EM, Bereczky GM. Liver necrosis from paracetamol. British journal of pharmacology and chemotherapy. 1966 Mar;26(3):606.
- Montgomery B. Does paracetamol cause hypertension?. Bmj. 2008 May 22;336(7654):1190-1.
- Turtle EJ, Dear JW, Webb DJ. A systematic review of the effect of paracetamol on blood pressure in hypertensive and non‐hypertensive subjects. British journal of clinical pharmacology. 2013 Jun;75(6):1396-405.
- Mehrotra TN, Gupta SK. Paracetamol-induced hemolytic anemia: report of a case. Indian journal of medical sciences. 1973 Jul;27(7):548-9.
- Gad SC. Cephalosporins. Encyclopedia of Toxicology: Third Edition. 2014
- Nath AP, Balasubramanian A, Ramalingam K. Cephalosporins: An imperative antibiotic over the generations. International Journal of Research in Pharmaceutical Sciences. 2020 Jan 20;11(1):623-9.
- Harrison CJ, Bratcher D. Cephalosporins: a review. Pediatrics in review. 2008 Aug 1;29(8):264.
- Malani PN. Mandell, Douglas, and Bennett’s principles and practice of infectious diseases. JAMA. 2010 Nov 10;304(18):2067-71.
- Thompson JW, Jacobs RF. Adverse effects of newer cephalosporins. Drug safety. 1993 Aug 1;9(2):132-42.
- Fonacier L, Hirschberg R, Gerson S. Adverse Drug Reactions to Cephalosporins in Hospitalized Patients with a History of Penicillin Allergy. In Allergy & Asthma Proceedings 2005 Mar 1 (Vol. 26, No. 2).
- Campagna JD, Bond MC, Schabelman E, Hayes BD. The use of cephalosporins in penicillin-allergic patients: a literature review. The Journal of emergency medicine. 2012 May 1;42(5):612-20.
- del Villar-Guerra P, Moreno Vicente-Arche B, Castrillo Bustamante S, Santana Rodriguez C. Anaphylactic reaction due to cefuroxime axetil: A rare cause of anaphylaxis. International journal of immunopathology and pharmacology. 2016 Dec;29(4):731-3.
- Sartori R, Colombo S. Stevens–Johnson syndrome/toxic epidermal necrolysis caused by cefadroxil in a cat. Journal of Feline Medicine and Surgery Open Reports. 2016 Jun;2(1):2055116916653616.
- Naranjo CA, Busto U, Sellers EM, Sandor P, Ruiz I, Roberts EA, et al. A method for estimating the probability of adverse drug reactions. Clinical Pharmacology & Therapeutics. 1981 Aug;30(2):239-45.
- Garcia-Cortes M, Lucena MI, Pachkoria K, Borraz Y, Hidalgo R, Andrade RJ, spanish group for the study of drug induced liver disease (grupo de estudio para las hepatopatías asociadas a medicamentos, geham). Evaluation of Naranjo Adverse Drug Reactions Probability Scale in causality assessment of drug‐induced liver injury. Alimentary pharmacology & therapeutics. 2008 May;27(9):780-9.
- Hartwig SC, Siegel J, Schneider PJ. Preventability and severity assessment in reporting adverse drug reactions. American journal of hospital pharmacy. 1992 Sep 1;49(9):2229-32.
- Lazarou J, Pomeranz BH, Corey PN. Incidence of adverse drug reactions in hospitalized patients: a meta-analysis of prospective studies. Jama. 1998 Apr 15;279(15):1200-5.
- Miguel A, Azevedo LF, Araújo M, Pereira AC. Frequency of adverse drug reactions in hospitalized patients: a systematic review and meta‐analysis. Pharmacoepidemiology and drug safety. 2012 Nov;21(11):1139-54.
- Bouvy JC, De Bruin ML, Koopmanschap MA. Epidemiology of adverse drug reactions in Europe: a review of recent observational studies. Drug safety. 2015 May 1;38(5):437-53.
- Sundaran S, Udayan A, Hareendranath K, Eliyas B, Ganesan B, Hassan A, et al. Study on the classification, causality, preventability and severity of adverse drug reaction using spontaneous reporting system in hospitalized patients. Pharmacy. 2018 Dec;6(4):108.
- Jha N, Bajracharya O, Namgyal T. Prevalence of adverse drug reactions with commonly prescribed drugs in different hospitals of Kathmandu valley. Kathmandu University medical journal (KUMJ). 2007;5(4):504-10.
- Pal SN, Duncombe C, Falzon D, Olsson S. WHO strategy for collecting safety data in public health programmes: complementing spontaneous reporting systems. Drug safety. 2013 Feb 1;36(2):75-81.
- Zopf Y, Rabe C, Neubert A, Gassmann KG, Rascher W, Hahn EG, et al. Women encounter ADRs more often than do men. European journal of clinical pharmacology. 2008 Oct 1;64(10):999.
- Lavan AH, Gallagher P. Predicting risk of adverse drug reactions in older adults. Therapeutic advances in drug safety. 2016 Feb;7(1):11-22.
- Pirmohamed M, James S, Meakin S, Green C, Scott AK, Walley TJ, et al Adverse drug reactions as cause of admission to hospital: prospective analysis of 18 820 patients. Bmj. 2004 Jul 1;329(7456):15-9.
- Beijer HJ, De Blaey CJ. Hospitalisations caused by adverse drug reactions (ADR): a meta-analysis of observational studies. Pharmacy World and Science. 2002 Apr 1;24(2):46-54.
- Gallagher RM, Kirkham JJ, Mason JR, Bird KA, Williamson PR, Nunn AJ, et al Development and inter-rater reliability of the Liverpool adverse drug reaction causality assessment tool. PloS one. 2011 Dec 14;6(12):e28096.
- Théophile H, André M, Miremont-Salamé G, Arimone Y, Bégaud B. Comparison of three methods (an updated logistic probabilistic method, the Naranjo and Liverpool algorithms) for the evaluation of routine pharmacovigilance case reports using consensual expert judgement as reference. Drug safety. 2013 Oct 1;36(10):1033-44.
Citation: Biswas S, Chatterjee A, Manna SSS, Kar U (2021) An observational study report on Adverse Drug Reactions of a combination of drugs in a tertiary care hospital, Kolkata, West Bengal, J. Pharamacovigil. 9:297. doi-10.35248/2329-6887.21.9.297
Copyright: © 2021 Biswas S et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

